All published articles of this journal are available on ScienceDirect.
Psoriasis Vulgaris: Relationship between Oral and Periodontal Conditions and Disease Severity
Abstract
Introduction:
Psoriasis has been associated with other diseases, or comorbidities, within which the oral cavity is involved. However, there is no consensus regarding the clinical description. The objective of this study was to establish the oral and periodontal status in a group of patients with Psoriasis vulgaris in relation to markers of clinical severity.
Materials and Methods:
Descriptive, cross-sectional, and observational study. 71 Patients diagnosed with psoriasis at the Hospital Militar Central. The indexes PASI and DLQI to determine their clinical activity were evaluated. We also evaluated serum markers of activity, A direct observation, ganglion palpation, extraoral, intraoral examination, and periodontal clinical index. Chi-squared test for associations was performed, All analyses involved a significance level of 5% and were carried out using the statistical analysis program STATA, version 11.1.
Results:
Periodontal disease was present in 70% of our patients with a moderate degree of severity in most cases. The presence of P. gingivalis was detected in 23% of patients, 9% of patients with P. gingivalis presented with severe psoriasis (p: 0.189); of these patients, 55% also had periodontal disease (p: 0.189); although this was not statistically significant. Furthermore, 91% of our patients presented with some form of lesion or anatomical variation in the oral cavity; of these, 28% had a single lesion, 63% had multiple lesions and 81.80% of the subjects showed lesions with strong psoriatic association (p = 0.033), including fissured tongue (60.87%; p = 0.034), angular cheilitis (14.49%; p = 0.03) and oral erythematous lesions (13.04%; p = 0.023).
Conclusion:
We did not detect statistically significant associations between the severity of psoriasis and periodontal disease but our findings related to oral changes could have good relevance for this type of patients. Our data indicate the need for multidisciplinary management between the clinician and specialists in periodontics and oral pathology.
1. INTRODUCTION
Psoriasis is distributed across the world with a prevalence ranging from 0.5% to 4.6% [1, 2]. This condition has been associated with other diseases, or comorbidities, such as oral cavity involvement, including oral lesions and periodontal disease, thus reaffirming that this is a systemic disease [3, 4]. Psoriasis is a chronic, inflammatory disease which is characterized by the exaggerated proliferation of keratinocytes as a result of immune system activation through T lymphocytes in focal cutaneous areas [5, 6].
In most cases of psoriasis, oral lesions are associated with the presence of geographic and fissured tongue, although data show an increased frequency of associations with generalized pustular psoriasis [7]. It has been estimated that the actual prevalence of fissured tongue and geographic tongue is 6 - 33% and 1-18%, respectively, suggesting that these might represent different expressions of the same disease [7]. However, there is no consensus of the clinical description of what could be considered an oral psoriatic lesion, oral evaluation, or examination, is not a regular procedure in patients with psoriasis [7, 8]. Oral lesions are generally asymptomatic, although there might be tongue swelling and pain when deep fissures are formed [7, 8]. Furthermore, bad tongue hygiene can cause halitosis and tissue swelling due to the accumulation of food residues in fissures, thus causing burning and stinging, especially after contact. However, this association is debatable because it is not known if such oral manifestations are a symptom of disease severity or represent an expression of the disease itself [8].
On the other hand, periodontal disease is marked by an exaggerated response of the immune system to oral microbiota, making it a chronic inflammatory disease which is mediated immunologically, in which immune cells cause inflammation and cellular destruction [9].
Although the two pathologies are similar from the immunological aspect, some characteristics still render them separate diseases and there is no real association known between them [10, 11]. New research has shown that both psoriasis and periodontal disease have both experienced increased prevalence worldwide, and have been associated with other diseases and comorbidities, thus reconfirming that these are, indeed, systemic diseases [12, 13]. There are no existing reports in the published literature which describe the prevalence and frequency of psoriasis and periodontal disease in Colombia. We considered that it was very important for such clinical data to be available because we believe that appropriate examinations should be carried out on all patients with psoriasis, who may have multiple associated comorbidities. We also believe that such examinations should be performed on a regular basis. Furthermore, it is not yet known if the diagnosis of oral psoriasis should be made when lesions in the oral cavity present by themselves, or only at the same time as skin symptoms develop. Consequently, in this study, we aimed to investigate the association between periodontal diagnosis, microbiological components, the presence of IgG against Porphyromonas gingivalis (P. gingivalis), and the clinical manifestations of psoriasis.
2. MATERIALS AND METHODS
2.1. Study Design
This was a cross-sectional descriptive study including patients who were clinically or histopathologically diagnosed with psoriasis. Clinical data were collected from all patients, analyzed and associated with serological test results. We then compared this information with that published in the existing literature.
2.2. Patient Population
The participants of this study were all patients of our dermatology department and were diagnosed with psoriasis between May 2014 and May 2015. The study was approved by an investigation committee and the Hospital Ethics Committee (Reference: HMC 2014-094) and was supported by Universidad Militar Nueva Granada (Code: MED-1800) the Hospital Militar Central and Universidad el Bosque.
We obtained 71 patients that fulfilled our inclusion criteria: over 18 years of age; diagnosis of psoriasis using the updated criteria of the 2012 Colombian Consensus of Psoriasis and the provision of informed consent [14]. For each patient, we used the Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index (DLQI) to determine clinical activity and impact on the quality of life [15, 16].
Patients with an active infection, malignancy, other autoimmune diseases, diabetes mellitus, antibiotic treatment over the previous three months, orthodontic pathology, and periodontal therapy within the last six months, pregnancy or lactation, were excluded from the study Table 1.
2.3. Periodontal/Oral Evalaution and Serum Markers
The periodontal evaluation included the determination of plaque and gingival indices, including bleeding to probing, loss of clinical insertion and the depth of probing. The quantitative polymerase chain reaction was carried out for P. gingivalis using primers and probes described by Boutaga et al. [17]. Indirect enzyme-linked immunosorbent assays were also used to investigate IgG1 and IgG2 antibodies against P. gingivalis [12]. We also determined Rheumatoid factor (447070, Immage 800, Beckman Coulter, Brea, CA), high sensitivity, C-Reactive Protein (CRP) (LKCRP, Immulite 1000, Siemens, Erlangen, Germany), and IgG/IgA ACPA (Quanta lite CCP 3.1 IgG/IgA), INOVA Diagnosis, San Diego, CA.).
Diagnosis of periodontal disease followed the guidelines established by the classification criteria of the Center of Control and Disease Prevention (CDC, USA) and the American Academy of Periodontics (AAP) [18]. Oral pathology examinations involved direct evaluation of the mucous membranes, inspection, and palpation of the lymph nodes and an extra-oral and intra-oral examination. Absolute and relative frequencies were established for oral, periodontal and microbiological diagnoses with central tendency scores and measures of dispersion for each clinical indicator. We then used the Chi-squared test to investigate for associations between the clinical and laboratory severity of psoriasis, periodontal and oral diagnosis, the presence of P. gingivalis and the serological levels of antibodies against P. gingivalis. All analyses involved a significance level of 5% and were carried out using the statistical analysis program STATA, version 11.1.
3. RESULTS
The mean age of our patients was 52.32±17.5 years, the mean measure of BMI was 26,41 ± 3,79 in the group. The distribution in separate groups by gender was the following: for men 27,33 ± 3,47 and 25,08 ± 3,9 for women (Table 1). Additionally, 62% of our patients had a body mass index (BMI) >25, and mean BMI was 26.4 ±3 kg/m2. Clinically, 97% of our patients had Psoriasis vulgaris (P. vulgaris) and 23% had nail lesions. Pitting was the most frequent presentation on the nail plate (15% of patients). Risk factor analysis showed that 46% of our patients had a history of smoking and 8% were active smokers.
| – |
Male n=42 |
Female n=29 |
Total n=71 |
|---|---|---|---|
| Mean±SD | |||
| AGE | 53,92 ± 16,5 | 50 ± 19,1 | 52.32± 17.5 |
| BMI | 27,33 ± 3,47 | 25,08 ± 3,9 | 26,41 ± 3,79 |
| n (%) | |||
| Comorbidity | 12 (44.44) | 15 (55.06) | 27 (38.03) |
| Current smoker | 1 (16.67) | 5 (83.33) | 6 (8.45) |
| History of smoking | 7 (21.21) | 26 (78.79) | 33 (46.48) |
| Economy | |||
| Home | 17 (100.00) | - | 17 (23.94) |
| Independent | 3 (33.33) | 6 (66.67) | 9 (12.68) |
| Employee | 2 (9.52) | 19 (90.48) | 21 (29.58) |
| Pensioner | 3 (15.00) | 17 (85.00) | 20 (28.17) |
| Student | 4 (100.00) | - | 4 (5.63) |
| Housing | |||
| Own | 20 (40.00) | 30 (60.00) | 50 (70.42) |
| Leased | 4 (33.33) | 8 (66.67) | 12 (16.90) |
| Common | 5 (71.43) | 2 (28.57) | 7 (9.86) |
| Accommodation | - | 2 (100.00) | 2 (2.82) |
| Marital status | |||
| Married | 17 (36.96) | 63.04(29) | 64.79 (46) |
| Single | 7 (53.85) | 46.15(6) | 18.31 (13) |
| Widower | 5 (71.43) | 28.57(2) | 9.86 (7) |
| Free union | - | 100.00(5) | 7.04 (5) |
| Degree level n (%) | |||
| Elementary school | 4 (44.44) | 5 (55.56) | 9 (12.68) |
| High school | 9 (33.33) | 18 (66.67) | 27 (38.03) |
| Technician | 8 (50.00) | 8 (50.00) | 16 (22.54) |
| University | 8 (42.11) | 11 (57.89) | 19 (26.76) |
BMI: Body Mass Index
3.1. Clinical Variables of Psoriasis
Clinically, 13% of patients had psoriatic arthritis, 97% had psoriasis vulgaris, and 41% presented with moderate to severe psoriatic activity. All patients were receiving topical corticosteroid therapy and 18% of patients were being treated with biological therapy. Methotrexate and anti-Tumor Necrosis Factor were the most commonly-used systemic treatments, with a frequency of 13% and 11%, respectively.
In terms of dermatology scales, the mean PASI score was 5.04±7.14, and was >10 in 14.08% of patients, while DLQI was >11 in 14.08% of patients. In total, 75% of patients with changes to the nails had severe psoriasis (p=0.002). Furthermore, 23% of patients had nail lesions; pitting was the most common type of lesion (15% of patients).
Plaque lesions on the head were found in 69 patients (97.18%), scaly lesions on the trunk in 48 patients (67.61%), with trunk extension in 70 patients (98.58%). Erythematous and scaly lesions on the upper limbs were observed in 46 individuals (64.79%) and compromised extension of the upper limbs was observed in 87.32% of patients. Erythematous lesions were evident on the lower limbs of 46 patients (64.79%; Table 2).
3.2. Laboratory Variables
None of the patients were positive for ACPA, with a mean level of 7.80± 0.07 U. Mean CRP level was 4.93±7.94 mg/L and mean Erythrocyte Sedimentation Rate (ESR) was 7.59± 8.75 mm3.
3.3. Oral Variables and their Association with Clinical Psoriatic Parameters
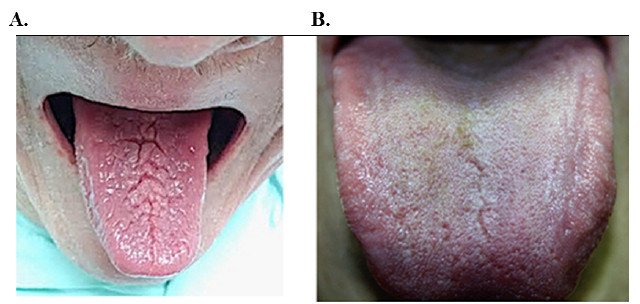
In total, 71 patients were analyzed; 91% of these patients presented with some form of lesion or anatomical variation in the oral cavity. Furthermore, 28% of patients had a single lesion and 63% presented with multiple oral lesions. Lesions associated with psoriasis were seen in 81.69% of patients, fissured tongue in 59.15% (Fig. 1A and 1B), followed by angular cheilitis 14.08%, erythematous lesions 12.67%, dry mouth (11.26%), oral burning (5.63%), geographic tongue (2.81%), and recurrent aphthae (1.40%) (Table 3).
| Dermatological Lesions | Total Psoriasis Group n=71 |
|---|---|
| - | n (%) |
| Evidence of erythematous lesion on head | 38(53.52) |
| Evidence of erythematous lesion on upper limbs | 46(64.79) |
| Evidence of erythematous lesion on lower limbs | 46(64.79) |
| Evidence of erythematous lesion on trunk |
43(60.57) |
| Evidence of scaly lesion on head | 35(49.30) |
| Evidence of scaly lesion on upper limbs | 46(64.79) |
| Evidence of scaly lesion on lower limbs | 43(60.57) |
| Evidence of scaly lesion on trunk | 48(67.61) |
| Evidence of indurated lesion on head | 32(45.07) |
| Evidence of indurated lesion on upper limbs | 45(63.38) |
| Evidence of indurated lesion on lower limbs | 42(59.15) |
| Evidence of indurated lesion on trunk | 44(61.97) |
| Head extension | |
| 0-10 | 69(97.18) |
| 10-30 | 2(2.82) |
| >30 | 0 |
| Upper limbs extension | |
| 0-10 | 62(87.32) |
| 10-30 | 2(2.82) |
| >30 | 7(8.86) |
| Lower limbs extension | |
| 0-10 | 26(36.62) |
| 10-30 | 45(63.38) |
| >30 | 0 |
| Trunk extensión | |
| 0-10 | 70(98.58) |
| 10-30 | 1(1.41) |
| >30 | |
| Evidence of nail lesions | |
| Presence | 16(22.54) |
| Nail lesion type | |
| Pitting | 11(15.26) |
| Drop of oil | 5(7.04) |
When analyzing the global severity indexes PASI and DLQI, no association was found with any oral lesion. However, analyzing the type of lesion individually, it was found that fissured tongue was significantly associated with the presence of comorbidities (p=0.034). The presence of angular cheilitis in 14.49% of patients was associated with ESR (p=0.03) and oral erythematous lesions, found in 13.04% of patients, were significantly associated with erythematous manifestations in the head (p=0.029), lower limb lesions (p=0.023), scaly head lesions (p=0.014), lower limb lesions (p=0.033), and indurated head lesions (p=0.043; Table 4). No other patient presented with actinic cheilitis or oral lichen planus. Overall, 42% of patients with oral lesions had severe psoriasis.
3.4. Periodontal Clinical Variables
Given our strict exclusion criteria, periodontal disease was evaluated only in 27 out of 71 patients. Our examinations showed that periodontal disease was present in 70% of patients, with a moderate severity in most cases (41%) (Table 5). P gingivalis was detected in 23% of these patients. More than half of the sample population (52%) had positive titers (titers >1/100) for IgG1 antibodies against P gingivalis and 37% for IgG2.
An association between the microbiological component and the presence of IgG against P gingivalis was evident, as well as the clinical variables of periodontal disease compared to the severity of the psoriatic disease, based on PASI score and treatment. We found that 65% and 62% of patients with IgG1 and IgG2 against P gingivalis, respectively, had mild psoriasis compared to those with severe psoriasis (p=0.362, p=0.489, respectively). Only 9% of patients with evidence of P gingivalis presented with severe psoriasis (p=0.189). Finally, 55% of patients with severe psoriasis had periodontal disease, although this association was not statistically significant (p=0.144).
| Oral Lesions | Total Psoriasis Group n=71 | |
|---|---|---|
| n (%) | ||
| Evidence of lesion associated with psoriasis | 62 (81.18) | |
| Evidence of fissured tongue | 42 (60.87) | |
| Evidence of angular cheilitis | 10 (14.49) | |
| Evidence of erythematous lesion | 9 (13.04) | |
| Evidence of dry mouth | 8 (11.59) | |
| Evidence of oral burning | 4 (5.80) | |
| Evidence of geographic tongue | 2 (2.90) | |
| Evidence of recurrent aphthae | 1 (1.45) | |
4. DISCUSSION
Psoriasis is an inflammatory disease that affects between 0.5 and 4.6% of the population worldwide, with rates varying between countries and races [1, 2, 6]. Evidence for oral lesions in psoriasis is rare and controversial, although some authors have reported non-specific lesions such as fissures and geographic tongue [7, 8]. This means that these conditions are oral manifestations of psoriasis because of their similarities in terms of clinical, genetic and histological findings. Nevertheless, there has been no conclusive statistical analysis which has investigated the association between periodontal conditions and the severity or clinical type of psoriasis [8].
Oral psoriasis manifestations were first described in 1903 [9], although its true association with the disease has been the source of much debate. Nowadays, there is evidence that oral lesions within a subgroup of patients with psoriasis are associated with skin lesions [7, 13]. Fissured and geographic tongue are considered to represent the most common oral manifestation in such patients [19] and a variety of studies have reported the prevalence of fissured and geographic tongue to be 35% and 17%, respectively [19, 20]. These findings could represent manifestations of P. vulgaris, thus acting as disease markers [21].
In 2009, a Brazilian study reported that geographic and fissured tongue were the most common oral lesions in patients with psoriasis [7]. The objective of the Brazilian study was to test if the frequency of oral lesions correlated with cutaneous psoriasis. A follow-up study was carried out on patients with P. vulgaris with geographic tongue to evaluate the progression of oral and cutaneous lesions. These lesions were more frequent in the psoriasis group and were not statistically associated with the frequency of oral lesions, although these types of lesions were significantly associated with the presence of cutaneous psoriasis [22].
In another study, Picciani studied 284 psoriatic patients to ascertain the relationship between severity and the presence of geographic tongue through PASI and demonstrated that severe psoriasis occurred in 25% of patients without geographic tongue and in 58% of patients with geographic tongue [23]. This author concluded that geographic tongue may be considered as a marker of severity [23].
In comparison, the results of our present showed that most patients had some type of oral lesion or anatomical variation, in addition to psoriatic-associated lesions, and that fissured tongue was the most common lesion associated with P. vulgaris. Our findings concurred with previous studies in that fissured and geographic tongue were clinical manifestations of P. vulgaris. Moreover, fissured tongue was more prevalent in late psoriasis while geographic tongue was more prevalent in early psoriasis, suggesting that these lesions can represent an oral manifestation of psoriasis.
As with our present study, previous literature, including a Venezuelan study by Kkilikan et al. [24] involving 200 patients with oral lesions, describes the analysis of different types of psoriasis without predilection for any form of the disease and reported that this type of oral manifestation has no specific association with any subtype of psoriasis, but does have an association with the serological marker, ESR, suggesting the need to evaluate the oral cavity in patients throughout the course of the disease. However, none of the existing studies had evaluated whether there is a direct association between other oral lesions and P. vulgaris or its severity. Since true psoriatic oral lesions and nonspecific changes are mostly asymptomatic, routine examination of the oral cavity seems necessary in all patients with the diagnosis of psoriasis [25, 26].
Another oral manifestation being investigated is the pre-sence of periodontal disease, which, like psoriasis, is showing increased prevalence worldwide [27]. However, there are very few existing reports that have confirmed the prevalence and prevalence and frequency collectively [28]. Prior to the present study, no study had evaluated the relationship between periodontal disease and psoriasis severity in Colombia. Both of these conditions are chronic inflammatory and destructive disorders, characterized by dysregulation of the host immune response. The etiology of both diseases is multifactorial and susceptibility is influenced by genetic and environmental factors [29]. Furthermore, changes in the microbiota of the gingival sulcus are a distinctive characteristic of the inflammatory hypothesis [30].
In 2016, the European Academy of Dermatology and Venereology [27] demonstrated an association between psoriasis and periodontal disease. Subsequently, a Danish study, involving a 15-year follow-up period, reported patients with mild to severe psoriasis and psoriatic arthritis [31]. The objective of this earlier study was to analyze the relationship between psoriasis or psoriatic arthritis and periodontal disease. Results showed that there was an increased risk of psoriatic patients developing the periodontal disease in comparison with the general population, and also that the incidence of periodontal disease was highest in patients with psoriasis [31]. The results of this previous paper concur well with our present study relating to P. vulgaris.
Our present data further showed that it was not possible to establish a significant association between the presence of P. gingivalis, antibody titer, and psoriasis severity. However, antibodies associated with other diseases, such as rheumatoid arthritis, have previously been shown to be associated with activity markers [12].
| Erythematous head lesion n (%) | P value | Erythematous trunk lesion n (%) | P value | Erythematous lower limb lesion n (%) | P value | Scaly head lesion n (%) | P value | Scaly lower limb lesion n (%) | P value | Indurated head lesion n (%) | P value | Indurated lower limb lesion n (%) | P value | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Erythematous lesion | 8 (88.8) | 0.029 | 7(77.78) | 0.305 | 9(100.00) | 0.023 | 8(88.89) | 0.014 | 9(100.00) | 0.033 | 7(77.7) | 0.043 | 9(100) | 0.033 |
| Geographic tongue | 1(50.0) | 0.884 | 1(50.00) | 0.715 | 1(50.00) | 0.612 | 1(50.00) | 0.983 | 1(50.0) | 0.542 | 1(50.0) | 0.917 | 1(50.0) | 0.542 |
| Fissured tongue | 23(54.75) | 0.948 | 27(64.2 | 0.674 | 29(69.0) | 0.601 | 21(50.0) | 0.881 | 30(71.4) | 0.675 | 19(45.0) | 0.813 | 30(71.4) | 0.675 |
| Angular cheilitis | 2(20.00) | 0.016 | 5(50.00) | 0.385 | 6(60.0) | 0.629 | 2(20.00) | 0.036 | 7(70.0) | 0.974 | 2(20.00) | 0.070 | 7(70.0) | 0.974 |
| Prosthetic stomatitis | 3(60.00) | 0.818 | 1(20.00) | 0.043 | 2(40.00) | 0.189 | 3(60.00) | 60.00 | 2(40.0) | 0.136 | 2(40.0) | 0.767 | 2(40.0) | 0.136 |
| Traumatic keratosis | 0(0.00) | 0.265 | 1(100.0) | 0.433 | 1(100.0) | 0.476 | 0(0.00) | 0.307 | 1(100.0) | 0.505 | 0(0.00) | 0.349 | 1(100.0) | 0.505 |
| Recurrent aphthae | 1(100.00) | 0.363 | 1(100.0) | 1.000 | 1(100.0) | 0.476 | 1(100.00) | 0.321 | 1(100.0) | 0.505 | 1(100.0) | 0.279 | 1(100.0) | 0.505 |
| Oral burning | 3(75.00) | 0.622 | 3(75.00) | 0.590 | 3(75.00) | 0.716 | 3(75.0) | 0.317 | 3(75.0) | 0.808 | 1(100.0) | 0.237 | 1(100.0) | 0.808 |
| Dry mouth | 4(50.00) | 0.759 | 5(75.00) | 0.991 | 6(75.00) | 0.595 | 7(87.5) | 0.965 | 7(87.5) | 0.241 | 2(25.00) | 0.197 | 7(87.5) | 0.241 |
| Periodontal disease | 10(52.63) | 0.637 | 9(56.25) | 0.053 | 11(57.8) | 0.401 | 11(57.8) | 0.637 | 11(57.8) | 0.401 | 9(47.37) | 0.472 | 11(57.8) | 0.401 |
| Parameters | Female | Male | Total |
|---|---|---|---|
| - | n=42 | n=29 | n=71 |
| Anti IgG1 P. gingivalis | - | - | - |
| 0-1/100 | 15 (44.12) | 19 (55.88) | 34 (50.00) |
| >1/100 | 14 (37.84) | 23 (62.16) | 34 (50.00) |
| Anti IgG2 P. gingivalis | - | - | - |
| 0-1/100 | 17 (37.78) | 28 (62.22) | 45 (63.38) |
| >1/100 | 12 (46.15) | 14 (53.85) | 26 (36.62) |
| P. gingivalis | - | - | - |
| Positive | 3 (50.00) | 3 (50.00) | 6 (23.08) |
| Periodontal disease diagnosis | - | - | - |
| Positive | 6 (31.58) | 13 (68.42) | 19 (70.37) |
| Periodontal disease severity | - | - | - |
| Low | 1 (25.00) | 3 (75.00) | 4 (14.81) |
| Medium | 3 (27.27) | 8 (72.73) | 11 (40.74) |
| Severe | 2 (50.00) | 2 (50.00) | 2 (14.81) |
No statistically significant differences
A significant frequency of periodontal disease was detected in patients with psoriasis in mild to severe forms, affecting more than half of the patient population sampled. Nevertheless, there was no statistically significant association in this respect, although this could have been due to the relatively small number of patients due to our strict exclusion criteria, some studies found a risk of periodontitis, Patients with the psoriatic disease should receive regular periodontal evaluation [32, 33].
Furthermore, the presence of lesions in the oral cavity, and its association with the severity of psoriasis indicates that in some patients, evaluation of the oral cavity should be considered as routine, especially given the possibility of using oral lesions as severity markers in some subgroups of patients with psoriasis, including P. vulgaris. This finding demonstrates the importance of oral health and the early detection of oral lesions because this practice might help control the disease in general.
Finally, our study was designed to raise awareness in the medical community of the potential effects of oral infection on systemic conditions. Our findings highlight the importance of multi-disciplinary management between dermatologists and periodontal and oral pathology specialists, in achieving an integral approach in medical practice, which would benefit patients and public health costs.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
All individuals who participated in the study signed informed consent.
HUMAN AND ANIMAL RIGHTS
No animals/humans were used for studies that are basis of this research.
CONSENT FOR PUBLICATION
Each participant signed an informed consent to participate in the study.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
This study was supported by Universidad Militar Nueva Granada code MED-1800 and Universidad El Bosque.
CONFLICTS OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
We appreciate the support provided by the central military hospital in the development of this study.


