All published articles of this journal are available on ScienceDirect.
Clinical Aspects of Atopic Dermatitis of Children in Brazzaville, Congo.
Abstract
Background:
Atopic dermatitis is a chronic inflammatory dermatosis for which racial specificities have been reported.
Objective:
To describe the clinical aspects of atopic dermatitis on children.
Methods:
Descriptive cross-sectional study in one public hospital and one private hospital in Brazzaville, Congo, for three years. Children under the age of 16, consulted for atopic dermatitis were included. Demographic and clinical data were collected. Atopic dermatitis was defined according to the criteria of the United Kingdom Working Party and its severity appreciated by SCORAD. The data was processed by EpiInfo 7® software.
Results:
Seventy-five children were included. They were 49 girls (65.33%) and 26 boys (34.67%), mean age 4.3 years [9 months-12 years]. Personal atopic history was found in 46.67% of cases and family atopic history in 61.33% of cases. Initial symptoms of atopic dermatitis had occurred before 2 years of age for 65 children (86.67%). The average consultation time was 33.4 months. In infancy, erythematous vesicular or oozing plaques were observed in 68.18% of cases and scaly erythematous plaques on folds in 50% of cases. In childhood, scaly erythematous plaques were observed in 84.90% of cases and lichenified eczema in 10.67% of cases. Xerosis was noted in 89.33% of all cases. Secondary infection was seen in 33.33% of cases. Twenty-five cases (33.33%) were severe.
Conclusion:
This study reveals a delayed diagnosis of atopic dermatitis, unusual clinical features and the importance of secondary infection. Information for families and clinicians should be considered to improve the earliness of the diagnosis.
1. INTRODUCTION
Atopic Dermatitis (AD) is a chronic pruriginous inflammatory dermatosis associated with atopy. It mostly begins during childhood, but can also begin in adulthood. The global prevalence of AD in children is 7.89%, with diversity by geographic area [1]. The disease has a heavy economic burden for the affected households, particularly in countries with low health coverage. It also has a significant impact on the quality of life of children and families [2,3].
The diagnosis of AD is based on clinical criteria. Clinical aspects have been widely described in the Caucasian population [4, 5], while on black skin, they remain insufficiently documented. As a result, racial and ethnic specificities regarding the clinical manifestations of AD are increasingly reported [6, 7].
In order to help define the patterns of atopic dermatitis in the black-skinned subject, the objective of this study was to describe the clinical aspects of atopic dermatitis on children in Brazzaville, Congo.
2. PATIENTS AND METHODS
This was a bicentric, descriptive cross-sectional study conducted from January 2016 to December 2018 in Brazzaville, Congo in two health facilities: Talangai Reference Hospital in Dermatology and infectious diseases Department and Cogemo private Hospital.
Children aged from 1 month to 15 years, consulted for atopic dermatitis in outpatient dermatology, were included. Children already treated for atopic dermatitis were not included. The diagnosis of atopic dermatitis was based on the criteria of the United Kindom Working Party [8].
The sampling was exhaustive in accordance with the inclusion criteria. The data were collected on a pre-established form during consultation through interrogation and physical examination. The variables studied were demographic data (age, sex, place of residence), history taken (personal and family history of atopic disease, age at the onset of symptoms, time interval and medication received before the consultation) and examination (type of dermatological lesions, topography, complications, degree of severity).
Among the complications, the bacterial secondary infection was defined by the presence of pus or honey-colored crusts on the lesions of atopic dermatitis and viral infection was defined by the presence of punched-out erosions or vesicles with or without fever [9, 10]. The degree of severity was appreciated by the SCORAD index [11, 12]. The AD has a score of 15 for mild cases, a score between 15-40 for moderate cases and above 40 for severe cases.
The data were processed using Epi Info7® software. The qualitative variables were expressed in number and percentage. Quantitative variables were expressed on mean with extreme values.
3. RESULTS
3.1. Socio-demographic Characteristics of the Study Population
Seventy-five children were included in the study. They were 49 girls (65.33%) and 26 boys (34.67%). The mean age was 4.3 years (9 months-12 years). Twenty-two children (29.33%) were under 2 years of age and 53 children (70.67%) were 2 years or older. Within all children, 34 (45.33%) were from a public hospital and 41 (54.67%) from a private hospital. The place of residence was urban for 74 (98.67%) children and rural for one child.
3.2. Clinical Aspects of Atopic Dermatitis
A personal history of atopy was noted in 35 cases (46.67%). These were allergic rhinitis in 20 cases (26.67%), asthma in 16 cases (21.33%) and allergic conjunctivitis in 8 cases (10.67%). A family history of atopy was found in 46 cases (61.33%).
Initial symptoms of AD had occurred before 2 years of age in 65 cases (86.67%), and after 2 years of age in 10 cases (13.33%). The average duration of the disease progression at the time of the consultation was 33.4 months (0-120). Prior to consultation, oral use of antibiotics was noted in 24 cases (32,00%). Local application of antibiotics, corticosteroids and vaseline was noted in 26 (34.67%), 30 (40.00%) and 23 (30.67%) cases, respectively. Five children had received traditional treatment.
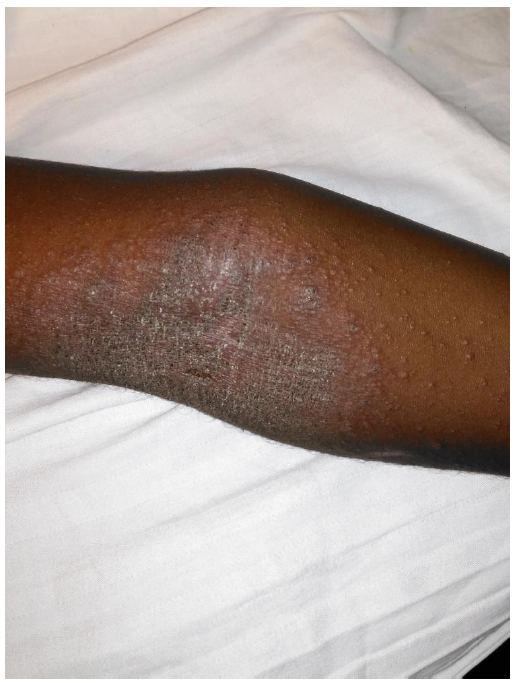
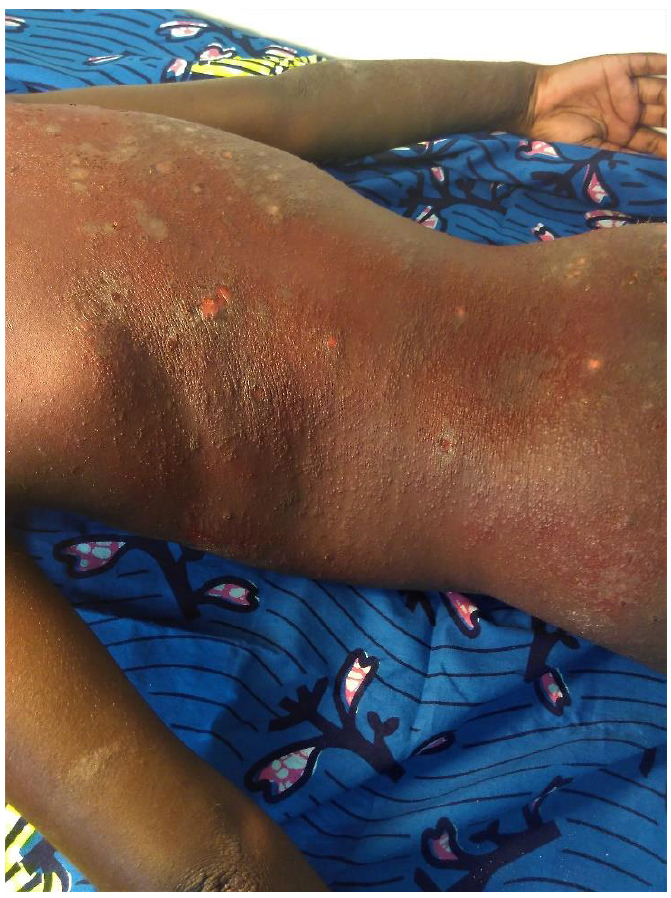
Pruritus was found in all children. Among the classic lesions of AD (eczema), erythematous vesicular or oozing plaques were found in 21 of 75 cases (28%), scaly erythematous plaques (Fig. 1) were found in 56 cases (74.67%) and poorly-demarcated papules (Fig. 2) were noted in 29 cases (68.67%).
The distribution of eczema lesions by age group is presented in Table 1.
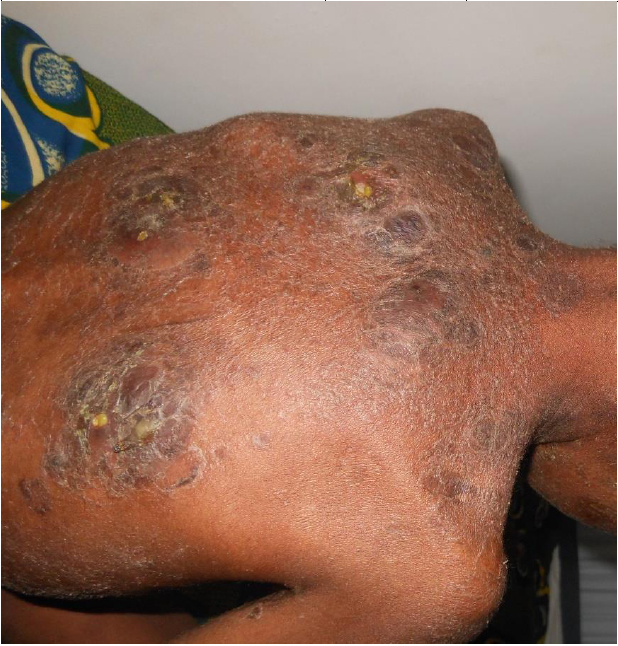
For atypical forms, the lichenoid atopic dermatitis (Fig. 3) was observed in 8 cases (10.67%), nummular-type atopic dermatitis in 7 cases (9.33%) and follicular atopic dermatitis in 4 cases (5.33%).
The various morphologic pattern might exist in the same child. The location of eczema lesions according to the clinical type and the age is presented in Table 2. The other clinical manifestations observed in children with atopic dermatitis, are reported in Table 3.
The degree of severity of atopic dermatitis was moderate in 50 cases (66.67%) and severe in 25 cases (33.33%).
Bacterial secondary infection was noted in 25 cases (33.33%) (Fig. 4).
4. DISCUSSION
The objective of our study was to describe the clinical aspects of atopic dermatitis in children, through a pediatric population consulted in outpatient dermatology.
The sample, composed mainly of children living in urban areas, is a limitation of this study. The diagnosis of AD was based on the United Kindom Working Party criteria because of its practical validity and high sensitivity [13-15]. These criteria are widely used for epidemiological and clinical studies of children [4].
Eczema is a cutaneous manifestation of atopy which is defined as a hereditary predisposition to develop IGE-mediated reactions to common environmental antigens [16]. A personal history of atopy was found in 46.67% of the children in our study and family history in 61.33% of cases. The frequency of atopy history varies depending on the studies. Out of a hospital study of 70 children in Côte d'Ivoire, Ahogo reports the existence of personal atopy in 57.24% of cases and a family history of atopy in 67.2% of cases [15]. In Israel, Rottem found a family history of atopy in 41.3% of cases in a hospital study [16]. In a community study in India, Narayan noted lower rates, 18.8% for personal atopy history, and 52.73% for family history [17].

| Clinical Characteristics of Eczema | < 2 years-old (infancy) N=22 |
≥ 2 years-old (childhood) N=53 |
||
|---|---|---|---|---|
| n | % | n | % | |
| Erythematous, vesicular or oozing plaques | 15 | 68.18 | 6 | 11.32 |
| Poorly-demarcated erythematous papules | 16 | 72.73 | 13 | 24.52 |
| Scaly erythematous plaques | 11 | 50.00 | 45 | 84.90 |
| Dermatological Lesions | Topography | < 2 years-old N= 22 Number (%) |
≥ 2 years-old N= 53 Number (%) |
|---|---|---|---|
| Erythematous vesicular or oozing lesions | Checks | 13 (59.09) | 4 (7.54) |
| Scalp | 1 (4.54) | - | |
| Neck | 1 (4.54) | - | |
| Folds | - | 2 (3.77) | |
| Scaly erythematous plaques | Large folds* | 11 (50.00) | 38 (71.69) |
| Trunk | - | 14 (26.41) | |
| Diffuse | - | 4 (7.54) | |
| Face | - | 4 (7.54) | |
| Neck | - | 8 (15.09) | |
| Thighs | - | 4 (7.54) | |
| Other locations | - | 15 (28.30) | |
| Pooly-demarcated erythematous papules | Face | 7 (31.81) | 7 (13.20) |
| Trunk | 7 (31.81) | 3 (5.66) | |
| Diffuse | 4 (18.18) | 2 (3.77) | |
| Other locations | - | 4 (7.54) | |
| Lichenified plaques | Extensor side of limbs | - | 7 (13.20) |
| Large folds* | - | 1 (1.88) | |
| Nummular patch | Trunk | - | 5 (9.43) |
| Legs | - | 2 (3.37) | |
| Follicular eczema | Trunk | - | 4 (7.54) |
| Clinical Features | Number | Percentage (%) |
|---|---|---|
| Xerosis | 67 | 89.33 |
| Keratosis pilaris | 11 | 14.67 |
| Retroauricular fissure | 6 | 8 |
| Pityriasis alba | 5 | 6.67 |
| Cheilitis | 5 | 6.67 |
| Dennie morgan infraorbital folds | 5 | 6.67 |
| Conjunctivitis | 5 | 6.67 |
| Prurigo papules | 2 | 2.66 |
Among the symptoms of atopy, the frequency of asthma in our study (21.33%) is close to Ahogo in Côte d’Ivoire (25.7%) [15], whereas a population study in Togo by Técléssou revealed asthma frequency of 12.08% [18]. The study by Técléssou also found an association between the personal history of asthma and the development of atopic dermatitis [18]. Different studies show that history of atopy is a diagnostic element whose association with atopic dermatitis varies according to studies: the history of atopy is generally lower in population studies than in hospital studies [19].
In spite of the age of onset of AD was before 2 years for 86.67% of the children in our study, access to care was late and the average consultation time was 33.4 months. Also, to help to treat symptoms, 32.00% of cases used oral antibiotics and 5 children resorted to inappropriate use of traditional medicine. Traditional treatments used on AD children were plant extracts or traditional oils. These practices have not been effective for the symptoms.
There are two major periods in the clinical manifestations of atopic dermatitis: infancy and childhood. During infancy, AD presents classically by acute eczematous lesions characterized by erythematous, vesicular papules found on the face, sparing the nose, and on the extensor side of extremities [20-22]. Flexural areas may be involved, particularly the neck fold [23]. During childhood, the involvement of the face is less frequent and eczema has a chronic presentation characterized by scaly erythematous or lichenified plaques of the large flexion folds and neck [5, 24].
Our study confirms the predominance of acute lesions localized on the cheeks, the trunk or diffuse. It reveals, in infants, the early onset of chronic eczema lesions on the large flexion folds without involving the limb extension faces. In children, the lesions observed in our study are similar to the common description of AD. However, the appearance of erythema and lichenifiction on black skin can make diagnosis difficult. The erythema is gray on black skin and the color is close to lichenification. The acute and chronic character of eczema lesions are often only distinguished by the thickening of the skin during lichenification. Atypical forms of eczema, such as follicular, nummular and lichenoid forms are classified as atypical forms of AD [20]. Among atypical forms of AD, the lichenoid form was found in 10.67% of cases and the follicular form in 5.33% of cases. Lichenoid atopic dermatitis and follicular AD are more frequent variants of AD seen in African-American and Latino-Hispanic patients [4, 7, 20]. Nnoruka, in Nigerian patients with AD, found a papular lichenoid lesion in 54% of cases [25]. Ahogo in Côte d'Ivoire observed this clinical form in 7.1% of cases [15]. Julian-Gonzalez in Mexico reports a lichenoid AD frequency of 7% [26]. A meta-analysis shows a high frequency of lichenoid forms in African countries, including Nigeria, Tunisie and South Africa [5]. Contrary to the usual localization, in our study, the lichenoid AD was located on the extensor side of the limbs or diffuse. Summey made the same finding on African-American with AD [27]. The lichenoid form is different from lichenification secondary to old lesions. AD lichenoid occurs early as flat lichen lesions, pigmented and sometimes hypertrophic. It requires a good appreciation of all diagnostic criteria of AD.
Xerosis was seen in 89.33% children. A meta-analysis showed that xerosis was among the top 5 most commonly reported features of AD in all regions of the world except Southeast Asia [5]. This analysis shows in 3 African countries, namely Nigeria, Tunisia and South Africa, that xerosis is found on average in 65% of cases [5]. The decreased and changed lipid content of the skin is one of the contributing factors to xerosis. Also, patients with AD have an increase in transepidermal water loss, which reduces the water content of the skin [28]. In our study pityriasis alba was found in 6.67% of AD children. It looked like hypopigmented patches located on the face and trunk. This condition is usually found at school age. Some studies report that this condition is more common in black-skinned subjects, but in fact, the condition is equi-revalent in all skin types, and just more apparent on the pigmented skin [29].
Among over the atopic stigmata, conjunctival disease, found in 6.67% of the children in this study, is lower than that seen by Carmi in France in 18% of cases [24]. In India, Kaujalgi [30] reports eye damage in 16% of cases and Sarkar [31] in 43% of cases.
In our study, 33.33% of cases of AD were complicated by a secondary bacterial infection. Ahogo in Côte d'Ivoire observed a lower frequency of 12.8% [15]. In the skin of patients with atopic dermatitis, malformation of the lipid layer, increased pH, and elevated transepidermal water loss contribute to the colonization and proliferation of S. aureus [32, 33].
In our context, other factors can contribute to the development of secondary infections. Delayed access to care is usually compensated by adverse therapeutic practices such as the application of inappropriate substances, which can alter the skin microbiota and promote infections. The unsanitary environment can also contribute to the infection.
CONCLUSION
Our study shows the early onset of chronic eczema form on large folds in infants, the predominance of lichenoid atopic dermatitis on extensor sides of extremities in children, and the importance of secondary infection. These findings should be taken into account to improve the diagnosis of atopic dermatitis
LIST OF ABBREVIATIONS
| AD | = Atopic Dermatitis |
| SCORAD | = Scoring of Atopic Dermatitis |
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
The Hospital Research Committee of Talangai Reference Hospital Brazzaville, Congo approved the study under the approval number 6947/MPPIFID /DGSSSa/DDSSSa-B/DST/HRT).
HUMAN AND ANIMAL RIGHTS
No Animals were used in this research. All human research procedures were followed in accordance with the ethical standards of the committee responsible for human experimentation (institutional and national), and with the Helsinki Declaration of 1975, as revised in 2013.
CONSENT FOR PUBLICATION
Informed consent was priorily obtained from all the parents.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


