All published articles of this journal are available on ScienceDirect.
Bullous Cutaneous Larva Migrans And Generalized Cutaneous Larva Migrans: A Rare Clinical Manifestation
Abstract
Background:
The skin lesions of Cutaneous Larva Migrans (CLM) commonly manifests as single, linear, irregular, serpiginous raised erythematous track, but the other clinical variants exist. This case series aimed to report one case of a vesiculobullous lesion in CLM and one case of CLM with generalized distribution serpiginous erythematous skin lesion.
Objectives:
We report one case of a vesiculobullous lesion in CLM and one case of CLM with generalized distribution serpiginous erythematous skin lesion.
Conclusion:
There were CLM with vesiculobullous lesions and also with generalized distribution of skin lesion. The pathogenesis of this condition is still unknown. In both cases, albendazole 400 mg per day for 3 and 5 days gave an effective result.
1. INTRODUCTION
Cutaneous Larva Migrans or sandworm eruption [1] is a creeping eruption caused by nematode larval migration in the epidermis [2]. The most common cause of this disease is Ancylostoma braziliense larvae, commonly found in dogs and cats [1, 3, 4].
Characteristics of early lesions in CLM are erythematous papules on the skin area where the larvae penetrated [3]. These skin lesions occur within one to five days after larval penetration [2, 3]. Furthermore, as the larvae migrate, developing tracks appear as a reddish raised linear line with a serpiginous pattern. Vesiculobullous lesions also can be found in 15% cases of CLM [3]. The skin lesions can be solitary or multiple or general, accompanied by itching. The most commonly affected body parts are the feet, hands, and buttocks [1]. There are 2 cases that reported a CLM case with severe clinical manifestations of multiple serpiginous skin eruptions [2, 3]. This CLM case series aimed to report a case with vesiculobullous lesions and a case with a generalized distribution of serpiginous erythematous skin lesions.
2. CASE 1
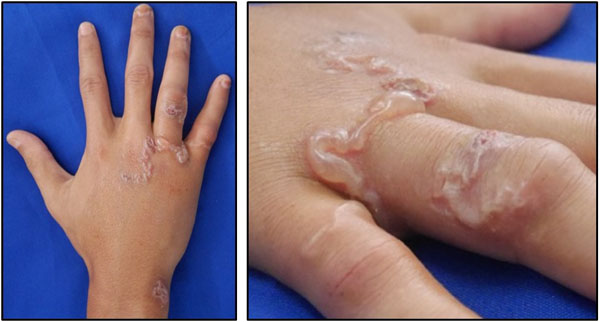
A 30-year old male came with bullous on the back of the wrist, 4th and 5th fingers of the right hand that feels itchy. He worked as a builder who often comes in contact with the soil and sand without using gloves. On physical examination, there were 7 cm bullous lesions with a serpiginous pattern (Fig. 1). Albendazole 400 mg daily was given for five days with good results.
3. CASE 2
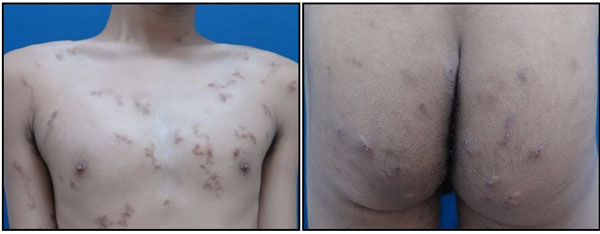
A 20-year old male came with erythematous papules and linear tracks in the entire body except for the face, hands, and feet that feel itchy. He had a history of traveling to the beach and buried his body into the sand. On physical examination, there were erythematous papules, erythematous, and hyperpigmented tracks in the serpiginous pattern, erosions, scales, hemorrhagic crusts, and hyperpigmented macules on the scalp, neck, trunk, buttocks, extremities, as well as the scrotum (Fig. 2). The patient was treated with albendazole 400 mg daily for three days and had a good result.
4. DISCUSSION
Cutaneous Larva Migrans is the most common helminthic infection of human skin and commonly seen in tropical [1] or subtropical countries, such as Southeast Asia, South America, and Africa [4]. This disease usually affects a person in contact with contaminated soil or sand and tourists [2] during a vacation at the beach [5].
The diagnosis of CLM can be established based on clinical manifestation [6-8] and supported by a history of contact with soil or sand during work or travel [6, 7]. At the penetration site of the larva, erythematous papules will develop after 1-5 days [9]. These skin lesions are accompanied by pruritus and it can be felt before the skin lesions are seen [7]. Larva migration along the skin creates a tract that appears as a reddish raised linear line with the serpiginous pattern [4, 5]. Tracts are formed between stratum basale and stratum granulosum [7, 9]. However, the larva can also persist for several days to several months before starting their migration. Linear lesions are often disconnected by the presence of papules that mark the location of the larvae [9, 10]. The size of tract varies with lengths of up to 15-20 cm and width of 3 mm [1, 11, 12]. The speed of larva migration is between few millimeters to 2 cm per day [2, 6]. The other CLM skin manifestations are vesicles or bullae [7, 10]. Vesiculobullous lesion can occur in 15% of CLM cases [1] Hochedez et al. [11] reported that vesiculobullous lesion could occur in 4-40% of CLM patients.
In general, most CLM patients have only a single larval tract. The skin lesions are commonly found in the lower extremities and buttocks, and only 7% of the CLM lesions manifest in the trunk and upper limb [12]. However, the skin lesions may occur in any location, including the face, scalp, and genital [7]. Sherman et al. [2] reported a CLM case in a 27-year-old male patient with severe manifestation after a beach trip. From the physical examination, it was found that there were multiple tracts with the serpiginous pattern on the buttocks and right limbs. Another severe CLM case was reported by French et al. [3] on an 18-year-old woman with multiple serpiginous tracts on chest and back. The presence of multiple larvae may cause multiple skin lesions [12].
In case 1, the patient worked as a builder who often came in contact with the soil and sand without using gloves. Initially, there were erythematous papules in the hands which then turned into vesicles and bullae accompanied by itching. The patient acknowledged that the lesions increased in length by about 1 cm per day. Based on physical examination, there were bullae in a serpiginous pattern with a length of 7 cm on the back of the wrist, 4th and 5th fingers of the right hand that feels itchy. In case 2, the patient with a history of traveling to the beach buried his body in the sand came with erythematous papules and linear tracks on the entire body except for the face, hands, and feet that feel itchy. Five days before the lesions appeared, he went to the beach and buried his body in the sand. During that time, he also denied eating raw fish. On physical examination, there were skin lesions with generalized distribution at scalp, neck, chest, abdomen, back, buttocks, arms, legs, and scrotum in the form of erythematous and hyperpigmented tracks, erosions, scales, hemorrhagic crust, and hyperpigmented macules.
Factors that cause vesiculobullous lesions and multiple lesions in CLM are still unknown. Shimogawara et al. [13] showed that there were increased levels of several cytokines, such as interleukin (IL)-4, IL-5, IL-6, and IL-10 in the peripheral circulation of CLM patients. The activation of the T helper-2 (Th2) cell leads to an increase in IL-5 level that causes eosinophil withdrawal [1] from the bone marrow to the peripheral circulation. Furthermore, IL-4 and other cytokines attract the eosinophils from the peripheral circulation to the epidermis to eliminate the larva [14].
In cases of CLM with severe symptoms, the patient can be given various treatments, either topical, systemic, or cryosurgery treatment [7]. Systemic treatment is the first-line treatment of CLM [15, 16]. Recommended treatment is the administration of systemic antihelmintic drugs, such as albendazole, ivermectin [1, 2], or thiabendazole [4]. Kincaid et al. [5] reported that over 90% of CLM cases with albendazole 400 mg per day for three days showed a good response. Albendazole can also be given up to seven days, especially in multiple skin lesions [17, 18]. Caumes et al. [19] suggested higher effectiveness of single-dose ivermectin compared to single-dose albendazole in CLM treatment. However, CLM with severe clinical manifestations, the administration of albendazole for three days gave a good result [12, 16].
In this case series, both patients were treated with albendazole 400 mg per day for three to five days. In case 1, albendazole 400 mg per day was given for five days. On the 5th day of treatment, there were no new skin lesions. In case 2, albendazole 400 mg per day was given for three days. On the 4th day of observation, itching was reduced, and there were no new skin lesions.
The skin lesions of CLM can disappear within 2-8 weeks [1, 17]. Both cases have improved skin lesions. Relapse in CLM can occur after 11 days of treatment with single-dose albendazole 400 mg, 16 whereas albendazole 400 mg per day for three days has efficacy of 100% [5]. In both cases, albendazole 400 mg per day was given for five and three days, respectively, without relapse.
CONCLUSION
CLM usually manifests as an erythematous, linear, irregular, serpiginous pattern tract, but other clinical variants can be found. There were CLM with vesiculobullous lesions and also with generalized distribution of skin lesion. That condition is supposed to be related to the immune response. In both cases, albendazole 400 mg per day for 3 and 5 days gave an effective result.
ETHICS APPROVAL AND CONSENT TO PARTICIPATE
This study was approved by the Health Research Ethics Committee of Dr Hasan Sadikin Hospital Bandung, Indonesia under approval number LB.02.01/X.6.5/277/2019.
HUMAN AND ANIMAL RIGHTS
Not applicable.
CONSENT FOR PUBLICATION
Each participant signed an informed consent to participate in the study.
STANDARD OF REPORTING
CARE guidelines and methodology were followed to conduct the study.
FUNDING
None.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.


