All published articles of this journal are available on ScienceDirect.
Proposal for a 4-type Classification of Acne: An Evidence-Based Review of the Literature
Abstract
Background:
Proper evidence-based classification and grading of a disease such as acne are important in guiding medical practitioners to properly diagnose diseases and treat patients.
Objective:
This is a review of the present classification of acne in order to delineate modified approaches of acne treatment.
Methods:
The available literature was reviewed, including searches from 7 databases based on the terms “classification of acne vulgaris and pathophysiology”, according to evidence-based medicine using the Cochrane risk of bias tool.
Results:
From a total of 10,121 studies on acne classification, 51 full-text articles were assessed and 13 studies were included after screening for acne classification.
Conclusion:
The European-evidence-based guideline (EDF) classification fits best. We propose a modified classification in 4 categories to improve the management of each stage of acne.
1. INTRODUCTION
A usable and practical classification or grading system of diseases is important both in medical practice and research. It allows medical practitioners to treat diseases in an appropriate and effective manner. A generally accepted grading system makes the comparison between different studies on the same disease much easier.
Acne vulgaris is a chronic inflammatory disease of the pilosebaceous unit and associated with serious consequences such as scar formation and a high psychological impact. There are multiple factors involved in the pathophysiology of acne vulgaris, including hormone-induced sebum production, follicular hyper-keratinization and Propionibacterium acnes (P. acnes) inducing inflammatory mechanisms (innate- and acquired immunity). P. acnes are Gram-positive anaerobic bacteria that are found abundantly in the pilo-sebaceous unit of the skin [1-3]. Epidermal cells, including keratinocytes, melanocytes and Langerhans cells, can recruit many inflammatory cells such as neutrophils, macrophages and natural killer cells via an activated innate immunity system, which explain that acne is an inflammatory disease [4]. Although many studies are performed in the last decades, acne is still a complex and sometimes difficult to treat disease.
Multiple grading systems for acne have been proposed by several groups of dermatologists since the 1930s. Among those grading systems, 2 main methods of assessment were used: 1. global severity grading and 2. lesion counting. The former is based on the overall appearance that is divided into different levels by comparison with the descriptive text or standardized photography. The latter classifies based on the number of each acne lesion and then multiplying the number of each type of lesion by a given severity index. Both systems were reported using numeric grading or sequential grading (mild, moderate, and severe) [5-7]. Proper classification will assist clinicians and patients in determining the severity of the disease and appropriate individual treatment [8].
The purpose of this review is to evaluate existing classifications of acne and how they can best relate to diagnosis and treatment.
2. OBJECTIVE
This study aims to review existing acne classification systems and their relationship with a rational therapeutic approach to diagnosis and treatment of acne vulgaris.
3. MATERIALS AND METHODS
3.1. Search Methods
The databases of PubMed, Embase, Scopus, Web of Science, Cochrane central registry of trials (CENTRAL) and Google Scholar were searched and documents collected until the end of September 2017. The search terms “acne vulgaris and classification and pathophysiology” were assessed using the Cochrane risk of bias tool [9]. The articles were selected by screening the title and the abstract. The full text of all the relevant articles was then assessed. The overall level of quality of evidence was assessed using the Grading of Recommendation, Assessment, Development and Evaluation (GRADE) approach [10].
4. RESULTS
4.1. Literature Search
The search found 10,121 articles (Web of sciences 58, Embase 389, PubMed 214, Medline (Ovid) 101, Cochrane 21, Scopus 168, Google Scholar 9,170 articles) for acne vulgaris classification and pathophysiology. 51 articles were assessed for the full- text. There were 13 full- text articles after screening by two independent investigators.
The exclusion criteria for full-text articles were articles on diseases other than acne vulgaris, not relevant, duplicate publication and publication in a language other than the English language. The details of the available classification are presented in Table 1.
5. DISCUSSION
From all acne classifications that were reported in the literature over the past 20 years, the best classification regarding the grade of evidence (grade A) by the Grading of Recommendation, Assessment, Development and Evaluation (GRADE) approach was the one provided by Zaenglein et al. [11], and European-evidence based guidelines (EDF guidelines) [12].
| Reference | Year | Grade of evidence | Classification/Grading of acne | Discussion |
|---|---|---|---|---|
| Zaenglein et al [11]. | 2016 | A | No universal acne grading/classifying system can be recommended | • Systematic review that was conducted by a multidisciplinary team, including 17 experts in the field of acne, 1 general practitioner, 1 pediatrician, and 1 patient • systematic search of evidence for 20 years and 242 abstracts were reviewed |
| Nast A. et al [12]. European evidence-base guideline |
2016 | A | Defined according to a global assessment • Comedonal acne: few comedones or no inflammatory lesions • Mild to moderate papulopustular acne (inflammatory lesions) • Severe papulopustular acne (inflammatory lesions) and moderate nodular acne (nodules or cysts) • severe nodular/conglobate acne(nodules or cysts) |
• Systematic review that was developed in accordance with the standard operating procedures of the European Dermatology Forum • systematic search of evidence of 5 years and results of 154 studies (28 from updated search) were compiled by the experts with regard to clinical relevance |
| Chee Leok GOH et al [13]. South-East Asia guideline |
2015 | C | Defined according to a global evaluation of the severity of acne lesions • Mild: few to several papules and pustules, no nodule • Moderate: several to many papules and pustules, few to many nodules • Severe: numerous or extensive papules and pustules, many nodules |
• 13 leading dermatologists from six countries in SEA (Hong Kong, India, Japan, Korea, Malaysia, the Philippines, Singapore, Taiwan, Thailand and the USA) adopt the ACC grading system for classification and selection of treatment for acne patients • simple to use |
| Dréno et al [14]. Global Evaluation Acne (GEA) scale |
2011 | C | Defined according to a global evaluation of the severity of acne lesions • Grade 0: Clear. No lesion • Grade 1: Almost clear. Almost no lesions (a few scattered open or closed comedones and very few papules) • Grade 2: Mild (easily recognizable: less than half of the face is involved. A few open or closed comedones and a few papules and pustules) • Grade 3: Moderate (more than half of the face is involved; many papules and pustules; many open or closed comedones; one nodule may be present) • Grade 4: Severe (entire face is involved, covered with many papules and pustules, open or closed comedones and rare nodules) • Grade 5: Very severe (highly inflammatory acne covering the face with the presence of nodules) |
• Developed by 7 expert dermatologists in the field of acne • small number of subjects (22 patients) • Difficult to recognize all the details of grading system • Training is needed for accurate grading |
| Hayashi et al. [24] | 2008 | C | Defined according to the number of inflammatory eruptions on half of the face • Mild: 0-5 • Moderate: 6-20 • Severe: 21–50 • Very severe: >50 |
• Evidence-based classification that was developed by groups of dermatologists from 13 Universities and 3 independent expert dermatologists in Japan • Large number of subjects (244 patients) • Using lesion counting, lesion identification and patient’s photography • Only inflammatory eruptions were involved in grading |
| Sinclair et al [15]. | 2005 | C | Defined according to the most severe lesions • Grade 1: Comedones only • Grade 2: Inflammatory papules present in addition to the comedones • Grade 3: Pustules present in addition to any of the above • Grade 4: Nodules, cysts, conglobate lesions or ulcers present in addition to any of the above |
• Consensus of a group of about 40 experts in the field of acne, mainly South African dermatologists • Sponsored by Galderma • No lesion count |
The latter classified acne in 4 categories; comedonal acne (open/closed comedones), mild to moderate papulopustular acne (superficial inflammatory lesions), severe papulopustular acne and moderate nodular acne, severe nodular/conglobate acne, which helped in the management of the acne correlated with the disease activity. The former concluded that no universal acne classification could be recommended. In this review, the European Dermatology Forum (EDF) guideline is considered as the most appropriate guideline for acne treatment among all existing classifications. However, it relies only on the overall severity of the disease. This classification is, therefore, not useful for grading individual lesions. Different lesions can be in a different stage of development of inflammation, which is very characteristic of acne. For research, the development of a single lesion during treatment is important, because different stages of inflammation could require different treatment options.
An international consensus from the Global Alliance points out that “There is no standardized acne grading or classification system” [16]. However, there is a dire need for a universal, easy and reliable classification system for acne. An appropriate treatment should be related to pathology, which is clinically seen based on the type of lesions. If the classification corresponds well to the underlining pathology, a treatment algorithm can be developed on the basis of the clinically obtained classification. Therefore, a classification based on the clinical presentation (of the type of acne lesions) with consequences of therapy would contribute to an ideal classification.
Acne vulgaris is well recognized as an androgen-dependent disease of the pilosebaceous unit of the skin [8]. The main factors involved in the pathogenesis of acne include hormonal involvement, abnormal keratinization, colonization of P. acnes, sebum production, and inflammatory events. All these factors are widely accepted as being involved in the pathogenesis of acne vulgaris. Finally, the clinical lesions are a reflection of inflammation; both the innate and the acquired immune system play an important role. The involvement of toll-like receptor 2(TLR2) in stimulating inflammatory pathways activation and inducing monocytes to produce IL-8 cytokine is through the peptidoglycan on gram-positive bacterial cell wall of the P. acnes [17]. Activation of NF-kβ through the myeloid differentiation protein (MyD88) associated kinase is the intracellular signaling pathway of TLR2 and TLR4 [4, 18]. The severity of inflammation may be related to the different types of P. acnes, as shown in the study by Dagnelie et al. [19]. This study presented evidence that helped conclude that P. acnes are a part of the normal flora on the skin and can activate the human innate immune system through TLRs by monocytes and keratinocytes. P. acnes can be divided into 6 phylotypes (IA1, IA2, IB, IC, II and III), whereby each different strain may induce a different immune response. They investigated patients with severe acne on the face and on the back and healthy controls. In patients with acne, phylotype IA1 was predominant (84.4%), especially in those with acne on the back (95.6%). In 71.4% of patients with severe acne, P. acne phylotypes were identical on the face and on the back, whereas this was the case in only 45.5% of the healthy controls. They reported that the severity of acne was associated with the loss of P. acnes phylotypes [19, 20].
Based on this review, the EDF guideline can be modified. We suggest a classification based on each stage of an acne lesion:
Acne type 1; skin color dome shape papule or comedone,
Acne type 2; moderate inflamed acne with faint red or pink papule,
Acne type 3; red papule, papulopustular lesion,
Acne type 4; nodule, nodulocystic, and cystic lesion. See Fig. 1. and Table 2.
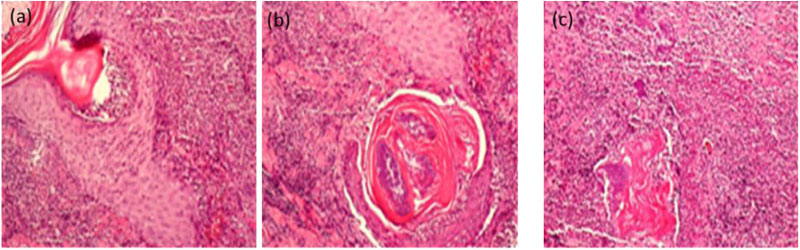
All clinical lesions of acne vulgaris are now considered inflammatory lesions, regardless of whether there is any clinical sign of inflammation [3]. The biopsy from the early stage of acne shows the feature of a lymphoid perivascular infiltrate. There is evidence that in the early stage of acne, lymphocytes and macrophages infiltrate hair follicle and release many inflammatory cytokines (IL-2, TNFα, IL-10, IGF1, TGFα, TGFβ, KGF, PDGF, etc.) [21]. Lytic enzymes and lipases released from P. acnes produce follicular epithelium disruption leading to inflammation of the acne [1]. In late stage acne, there is a neutrophilic response and myeloid cells phagocytize P. acnes in the dermis via the chemotactic factors [1, 22]. Polymorphonuclear leukocytes (PMN) infiltrate in the lesion, lead to pustule formation and cause rupture of the lesion [3, 4]. The inflammatory cytokines cause vasodilation of vessels and melanogenesis [21]. Histologically, there are differences in the cell types that are located at different depths and also different sites and different severity of ruptures of the affected infundibular wall as shown in Fig. (2).
The appropriate clinical criteria should start from the sequence of acne development. The stage of acne starts from mild to moderate and severe inflammation. Mild and moderate lesions usually resolve without any consequences such as post-inflammatory erythema or hyperpigmentation. While a severe acne lesion almost always is followed by a complication. Acne guideline recommends to scale the acne lesions of the whole face, applying every anti-acne product on the whole face or prescribing the systemic medication. The research and practice from Linda S Gold et al. [23], addressed that “it is very uncommon for inflammatory and non-inflammatory lesions to respond to an acne therapy in the same fashion over the same timeframe “. We have noticed that each type of lesion is not only clinically different but also there are differences and similarities in their histology, immuno histochemistry of the inflammatory cell infiltration, the wall of the pilosebaceous apparatus and inflammatory reaction, as summarized in Table 3. So they may share the same kind of therapy and they should be treated differently dependable on future discovery.
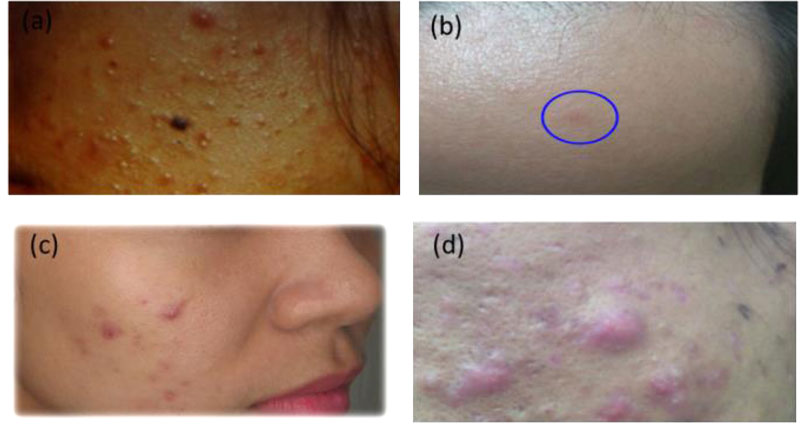
Acne type 1, mildly inflamed papular lesions
Acne type 2, moderately inflamed, pink papules
Acne type 3, fully blown inflamed, red papulopustules
Acne type 4, deep-seated papules or nodulocystic.
| New Classfication | Clinical Presentation | European Evidence-Based Guideline (EDF) |
|---|---|---|
| Acne type 1 | skin color dome shape papule or comedone | Comedonal acne |
| Acne type 2 | moderate inflamed acne with faint red or pink papule | Mild to moderate papulopustular acne |
| Acne type 3 | red papule, papulopustular lesion | Severe papulopustular acne and moderate nodular acne |
| Acne type 4 | nodule, nodulocystic, and cystic lesion | severe nodular/conglobate acne |
| Type of Lesion | Histology / Immunohistochemistry | Type of Inflammation | Therapeutic Rationales | |||
|---|---|---|---|---|---|---|
| Cell Inflammation | Pilosebaceous Apparatus | TLR2 | TLR4 | Major Rx | Adjunctive Rx | |
| Type 1 mild | Lymphocyte no neutrophil |
Keratin plugging (Comedone) |
Positive | Mostly negative | Comedone extraction | Anti TLR2 |
| Type 2 moderate | Lymphocyte and monocyte no neutrophil |
Minor rupture of upper part | Positive | Mostly negative | Anti TLR2 | - |
| Type 3 severe | Neutrophil predominate | Major rupture of upper part | Positive | Mostly negative | Anti TLR2 | Enhance wound healing process |
| Type 4 very severe | Neutrophil predominate | Major rupture at lower part | Positive | Positive | Anti TLR4 | • Enhance wound healing process • Anti TLR2 |
CONCLUSION
There are multiple favorable acne classifications; this evidence-based search gives an advice that among all the classifications, the EDF fits best. However, none of them classified acne by each individual stage of acne lesions and its root cause. The suggested improved classification should be able to combine clinical, molecular biological and histological features, which may lead to improved identification and management of each stage of acne.
LIST OF ABBREVIATION
| CENTRAL | = CENTRAL Cochrane central registry of trial |
| EDF guidelines | = European-evidence based guidelines |
| GEA | = Global Evaluation Acne scale |
| GRADE | = Grading of Recommendation, Assessment, Development and Evaluation |
| MyD88 | = Myeloid differentiation protein |
| P. acnes | = Propionibacterium acnes |
| TLR2 | = Toll-like receptor 2 |
| TLR4 | = Toll-like receptor 4 |
CONSENT FOR PUBLICATION
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The author declares no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
I would like to express my deep gratitude to Dr. Wanvipa Thongborisute for her assistance in searching and reading articles. I would like to express my very great appreciation to Dr. Suthep Jirasuthat for his advice and for performing histopathology.


