All published articles of this journal are available on ScienceDirect.
The Benefits Of Occlusive Dressings In Wound Healing
Abstract
There are several types of wounds with their own healing properties. The latest innovation in wound management by using occlusive dressings can prevent infections, improve healing time and patient’s comfort. Occlusive dressings are often used as an immediate wound hygiene control and also prevent blood loss until debridement is performed. They are used to protect wounds and surrounding tissue from pathogens and other harmful materials. A good cover depends on the condition around the wound, the person's skills, and the injury's nature. In this article, we provide an insight into the types of polymer materials used clinically in wound dressing and underlying mechanisms between the biomaterial dressings and the body tissue.
1. INTRODUCTION
In general, wounds can be classified into acute and chronic wounds. Acute wounds mostly tend to heal completely within 8–12 weeks with minimal scarring. Chronic wounds tend to reoccur and have a prolonged healing time that extends beyond 12 weeks. Wounds can also be classified according to skin layers and affected areas. Superficial wounds involve only the epidermal skin surface. Partial thickness wounds involve the epidermis, deeper dermal layers, blood vessels, sweat glands, and hair follicles. Full thickness wounds are caused by injuries that extend into the subcutaneous fat or deeper tissue along with the epidermis and dermal layers [1, 2].
In 1958, it was the first time known that moist environment for the wound could improve wound healing, when it was found that blisters healed faster if left in their original form [3]. Previously, honey paste, plant fiber, and animal fat were commonly used. Gauzes, lint, and cotton wool began to be used as wound dressings a few years later [4]. It was found in several studies that a polyethylene film made partial-thickness wounds of porcine heal approximately two times faster than the wound left open to the air. After that, many studies agreed about the benefits of moist wound environment for acute wound healing [3]. This has been found in acute but not chronic wounds because no animal model exists for chronic wounds. Due to excessive wound drainage from the traditional dressings, it becomes moistened and tends to become adherent to the wound making it painful when removing. These traditional dressings fail to provide moist environment to the wound so they have been replaced by modern dressings with more advanced formulations [5]. Moist condition makes wound healing faster, relieves pain, prevents infection and contamination than acute wound treated with other methods [3, 6]. To prevent infections, occlusive dressings need to be changed regulary. Occlusive dressings are mostly produced in the form of a flat shape sheet. Some of them can be cut according to the size of the wound, but some of them also need secondary dressings to completely seal the wound [7].
2. WOUND HEALING
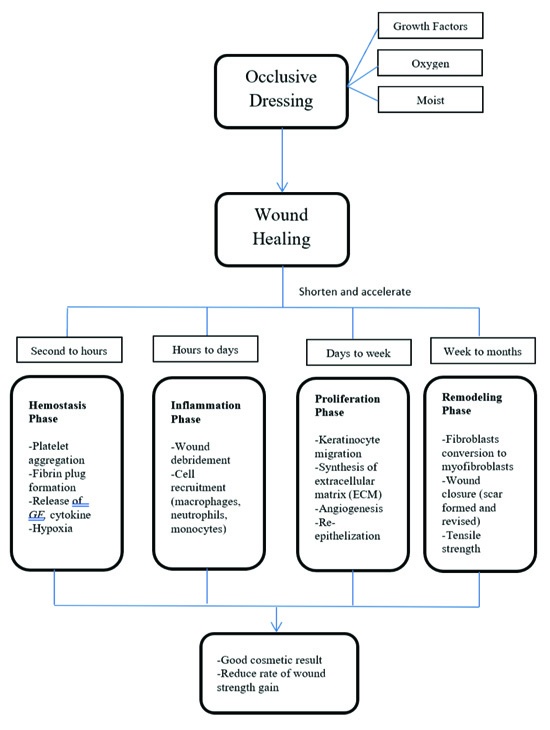
Wound healing is a complex proccess which is influenced by various mechanisms involving the coordinated interaction of blood cells, proteins, proteases, growth factors, and extracellular matrix components. Wound healing process can be divided into four phases (1) the coagulation and haemostasis phase; (2) inflammatory phase; (3) proliferative phase; and (4) maturational phase [8].
Hemostasis is initiated during the exposure of collagen during wound formation that activates the intrinsic and extrinsic clotting cascade. Injury to tissue causes a release of thromboxane A2 and prostaglandin 2-alpha which leads to vasoconstriction. Extravasation of blood constituents assists in the formation of the blood clot. All of this process helps to limit hemorrhage and provides an initial extracellular matrix for cell migration.
After hemostasis is achieved, inflammatory phase begins. Capillary vasodilatation and leakage result secondary to local histamine release by the activated complement cascade. The increased blood flow and altered vascular permeability allow for the migration of inflammatory cells to the wound bed. Complement, neutrophil and macrophage play an important role in stimulating inflammatory cells, bacterial lysis and debris scavenging. In addition to direct phagocytosis of bacteria and foreign materials, macrophages secrete various enzymes and growth factors.
The proliferative phase is marked by epithelialization, angiogenesis, granulation tissue formation, and collagen deposition. Epithelialization occurs within hours after injury in wound healing and the normal layers of epidermis are restored typically in 3 days. The proliferative phase ends with granulation tissue formation.
The maturational phase is characterized by transition from the granulation tissue to scar formation. Around two weeks after injury, the wound undergoes contraction which results in a smaller amount of apparent scar tissue. Collagen deposition by fibroblasts continues for a prolonged period with a net increase in collagen deposition reaching after three weeks from tissue injury.The entire process is a dynamic continuum dictated by numerous growth factors and cells with an overlap of each of the three phases of wound healing to provide continued remodeling.The human wound has its maximal strength at one year [4, 9].
Promotion of these phases largely depends on the wound type, its associated pathological conditions and also the type of dressing material to achieve faster healing. Several factors can impair the whole process of wound healing. In general, these factors are classified as local and systemic. Local factors that affect wound healing are desiccation, infection, maceration, necrosis, oxygenation, pressure, trauma, and edema. (Checklist wound healing). Systemic factors such as aging, hormones, stress, and systemic diseases are essential in determining individual wound healing process [8, 10-14].
3. BASICS OF OCCLUSIVE DRESSINGS
Based on the wound type, suitable dressing material must be used. Dressing selection should be based on its ability to a) provide or maintain moist environment, b) enhance epidermal migration, c) promote angiogenesis and connective tissue synthesis, d) allow gas exchange between wounded tissue and environment, e) maintain appropriate tissue temperature to improve the blood flow to the wound bed and enhance epidermal migration, f) provide protection against bacterial infection, g) should be non-adherent to the wound and easy to remove after healing, h) must provide debridement action to enhance leucocytes migration and support the accumulation of enzyme, and i) must be sterile, non-toxic and non-allergic [15].
Modern wound dressings are usually based on synthetic polymers and are classified as passive, interactive and bioactive products. Passive products are non-occlusive, such as gauze and tulle dressings, used to cover the wound to restore its function underneath. Interactive dressings are semi-occlusive or occlusive, available in the form of films, foam, hydrogel and hydrocolloids. These dressings act as a barrier against penetration of bacteria to the wound environment [16-19].
Occlusive dressings increase about 40% of the process of re-epithelialization of partial thickness wound [4, 20]. Studies where occlusive dressing leads to earlier epithelization during wound healing have been proposed [20]. When the wound is closed with dressing, they are continuously exposed to proteinases, chemotactic, complement & growth factors, which is lost in the wound exposed. These dressings help in faster re-epithelialization, collagen synthesis, promote angiogenesis by creating hypoxia to the wound bed and decrease wound bed pH which leads to a decrease in the wound infection [21]. An increase in oxygen tension under occlusion has also been suggested as a beneficial effect of these dressings; however, hydrocolloids, which have healing rates as good as or better than those of polymer films, are actually oxygen impermeable, whereas the films are oxygen permeable.
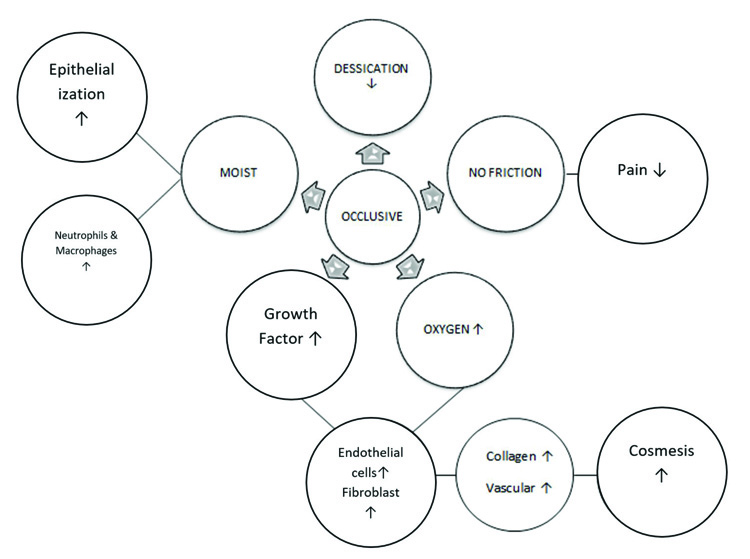
Inflammatory phase and angiogenesis are enhanced in moist wound environment when compared to dry wound environment. Based on Winter et al. studies, dry wound tends to heal slowly with poor cosmesis compared with wound in a moist environment due to impaired reepithelialisation and scab formation [16, 19]. Fig. (1) describes the basic mechanism of occlusive dressings.
The number of endothelial and fibroblast cells increases in moist wound conditions as compared to wounds that are kept dry. Inflammatory phase cells such as neutrophils and macrophages increase in moist wound environment which can reduce scab or debris formation. By preventing wound desiccation, occlusion maintains an electrical gradient between wounded and non-wounded skin, which may stimulate epidermal cell migration.
Fluid in the wound improves wound healing by interacting with regenerating epithelium, dermal, and granulation tissue (ie, fibroblasts and endothelial cells). An increase in oxygen tension in combination with electrical stimulation has been shown to increase the expression of growth factor receptors on endothelial cells and fibroblast. Endothelial cells and fibroblast stimulated by platelet-derived growth factor increase collagen synthesis and promote angiogenesis. This mechanism plays an important role in preserving cosmesis. Inflammatory infiltrate develops from the dominant neutrophils to macrophages in mature wounds. Occlusion shortens the inflammatory healing phase and produces good cosmetic results. Fig. (2) describes the effects of occlusive dressings in wound healing process [4, 5].
It was thought that occlusion of wounds may lead to an increased risk of bacterial infection, but studies showed that wounds treated with dressings, which promote moist wound environment, are associated with a lower infection rate. Dermatitis has been reported after prolonged exposure to water, but such hydration induced changes to the epidermis can be reversed quickly if the underlying cause is removed. A wound dressing thus approaching ideal characteristics should conform to the site of the wound, offer alleviation of pain symptoms, promote faster wound-healing time and attempt to restore the patients’ normal daily activities. Occlusive dressings protect the wound from both pathogenic invasion and further trauma. They act as barriers and prevent the entry of outside pathogens that might infect the wound and retard healing. Since they conform to the body's contours, they are more likely to remain in place and offer protection from further trauma as well [4, 22-26].
4. INDICATIONS AND CONTRAINDICATIONS FOR OCCLUSIVE DRESSINGS
We summarized the indications and contraindications of occlusive dressings and also its beneficial effects.
Indications of occlusive dressings are [7]:
- Wound treatment. Occlusive dressings can be comb- ined with antibiotics, gauze, sponge, hydrogels, and any other method to treat wounds
- Pressure and bleeding can often be immediately addressed
- Atopical ointments. It gives pressure to the ointment which improves absorption into the wound and prevents evaporation.
- Partial evisceration. Occlusive dressings are applied to protect the bowel until surgery.
- Sucking chest wounds. Wound and the puncture are covered and treated.
Contraindications of occlusive dressings are [7]:
- Maceration of the skin, where the skin feels tender, moist, and looks whiter than before, is an occlusive dressing's long-term risk.
- Pathogenic germs that exist in the infected area.
Benefits of occlusive dressings [6]:
- Improve patient’s comfort
- Lower the risk of infection
- Speed up wound healing
Several studies show the benefits of occlusive dressing for treating herpes virus skin infection. Based on histological analysis, it is known that skin injury caused by herpes virus is similar to partial-thickness skin wounds. Based on the study by Keegan et al., occlusive dressing relieved pain and hypersensitivity caused by herpes zoster infection which may lead as an option to rule out the need for oral analgesics. A blinded control study by Lin et al. showed that the use of plain Tegaderm™ provided better pain relief. Lee et al. also showed the benefits of occlusive dressing for the management of herpes zoster by improving wound healing, relieving pain, improving patient’s comfort and eschar debridement [27-29].
5. TYPES OF OCCLUSIVE DRESSINGS
Occlusive dressings work by maintaining moist wound environment which can shield the wound surface by preventing dry environment and trauma. Dry environment can prevent new epidermal cells migration to the wound surface. Variant types are: 1) semipermeable films, 2) hydrogels, 3) hydrocolloids and 4) alginates. Examples, functions, and features of each follow [6].
5.1. Polymer Films
In 1960s, film dressings were introduced for wound management. These transparent semipermeable dressings are made of polyethylene or polyurethane with an adhesive coating on one side, are thin, highly elastomeric, elastic, permeable to moisture vapor and atmospheric gases but impermeable to liquid and bacteria [3]. Originally, these films are used for healing minor burns, superficial wounds, superficial decubitus ulcers, donor sites, blisters, abrasions, and cuts [6].
During the past years, wound dressing has been modernized which now mainly consists of synthetic polymers for wound management and can be classified as passive and interactive. Passive synthetic polymer dressings are non-occlusive, used for covering wound and help in restoring function under the polymer film. Examples of such passive synthetic polymers are gauze and tulle. Interactive synthetic polymer dressings are occlusive or semi-occlusive which provide a barrier against bacterial penetration to the wound [30].
Polymer films trap exudates so the wound environment becomes moist [3, 4]. One main feature is that these films are impermeable to bacteria and liquid but are permeable to moisture vapor and air. The major drawback of polymer dressings is that exudates from wounds may accumulate underneath these dressings, which, due to increased pressure may cause a break in the environment maintained by the occlusive dressing [31, 32]. These products can be used for long-term bedridden patients acting as a shield to protect against skin friction in vulnerable areas, as a cover over topical products, and as intravenous dressings [3, 6].
5.2. Polymer Foams
Polyurethane foam film dressing is made of a thin microporous sheet of polyurethane foam that has been coated with a hydrophilic adhesive and bonded to a polyurethane film layer, which is good to avoid dehydration and bacterial infection. Foam film dressing has small pore size which increases the effectiveness of absorption or fluid retention. It also provides effectiveness in the prevention of bacterial infection and dehydration. Foam dressings attempt to rectify the lack of exudate absorbancy of occlusive dressing, without compromising on the moist environment needed for tissue repair [33-36]. Foams dressing does not cause maceration even if used for several days [4]
The major weakness of this type of dressing is that it needs secondary dressing like elastic bandage or a film to attach to the wound. Foam dressings are used for the wound that produced many exudates and deep wound or used under the compression bandage for chronic wound like venous ulcer [3, 4].
5.3. Hydrogel Dressings
Hydrogel contains about 96% water but does not dissolve in water and causes swelling if exposed to liquid solutions which can transmit moisture vapor. The high water content of hydrogels helps granulation tissues and epithelium in a moist environment. Hydrogel is made of polymers (polyethylene oxide, polyacrylamide, and polyvinylpyrrolidone) which are insoluble hydrophilic. Soft elastic property of hydrogels provides easy application and removal after the wound is healed without any damage. Their rate of absorbance is low, but high in capacity. For application, hydrogels need a secondary layer to attach to the wound, such as bandage or tape.3 Hydrogel has soothing and cooling effect up to 5°C and maintains reduction in the cutaneous wounds temperature for up to 6 hours (may be augmented by refrigeration before use) which can alleviate pain and inflammation.
| Type | Purpose Use For |
|---|---|
| Polymeric hydrogels | Ulcers Chemotherapy peels Laser resurfacing |
| Polymeric foam | Burns Chronic wounds Laser resurfacing wounds Mohs surgery and wounds |
| Polymeric hydrocolloides | Average thickness wounds Burns Chronic Ulcer |
| Polymeric alginates | Surgical wounds Thickness burns Chronic ulcer High exudate wounds |
The main disadvantage for using hydrogel is that it is easy to dry out which reduces their effectiveness and needs to be changed after 2 days [6]. Hydrogel is also a bad bacterial barrier.3 Morgan has reported that except for infected and heavy drainage wounds, hydrogel dressings are suitable for all four stages of wound healing. Difficulties in hydrogel dressings are that exudate accumulation leads to maceration and bacterial proliferation that produces foul smell in wounds. Besides, low mechanical strength of hydrogels makes it difficult to handle [36, 37]. Hydrogel is indicated for partial-thickness burns, dry chronic wounds, necrotic wounds, blisters and minor lacerations (Table 1) [3].
5.4. Hydrocolloid Dressings
Hydrocolloid is waterproof, semipermeable to vapor, opaque, occlusive, and absorbent. Hydrocolloid can be obtained synthetically or naturally [4]. It consists of two layers, inner colloidal layer and outer water-impermeable layer [3]. Hydrocolloids often use polysaccharides gel agents (sodium carboxymethyl cellulose), elastomeric compound, and are adhesive [4]. This type of dressing does not need secondary dressing and is impermeable to gases and moisture vapor [1]. Hydrocolloids are permeable to water vapor but impermeable to bacteria and also have the properties of debridement and absorb wound exudates [38]. They can melt so when they absorb exudate, but become viscous gel to stay in the wound when the dressings are removed. Hydrocolloids can be removed by normal saline [3].
The encapsulation properties of hydrocolloids create an opportunity to produce enhanced products with controlled drug (or any other appropriate substance) release due to the different type of hydrocolloid gels with different structural gelation. The amount of drug release can be controlled by optimization of the size of particles and the permeability of the gel membrane [39, 40].
Hydrocolloid dressings are designed on some wounds for prolonged periods (more than 1 week); this is useful in managing clean ulcers, but not when regular wound inspection is required. Thus, these dressings are probably more useful in preventing, rather than treating, infection within a wound [41]. The disadvantages of this dressing are the “gel and smell” phenomenon which may create an odorous residue when the exudate is mixed with adhesive when the dressing is removed.6 In medical applications, the current market for hydrocolloid dressings offers their use in healing diabetic foot ulcers, chronic wound management, burn, and are also recommended for paediatric wound care management, as they do not cause pain on removal [3, 4].
5.5. Alginates
Alginate dressings are produced from the calcium and sodium salts of alginic acid which can be obtained from seaweed. Alginate dressings can be in the form of nonwoven mats, porous (foam) sheets or twisted staple fibers.3 Alginate works by ion exchange reaction. The dressings would swell when wound exudates or sodium ions in tissue fluid are exchanged with alginates dressings of calcium ions. Alginate forms a gel in contact with the exudates. This property of alginate gels makes them very desirable as dressing for wounds that contain low to high exudates [42]. It would stay as gel for around one month if compared to hydrocolloids which degrade faster [3, 4].
| Polymer Foams | Hydrogels | Polymer Films | Alginates | Hydrocolloids | |
|---|---|---|---|---|---|
| Absorbent | + | + | - | + | ± |
| Transparant | - | ± | + | - | - |
| Suited for exudative wounds | + | ± | - | + | - |
| Permeable to water vapor | + | + | + | + | - |
| Permeable to oxygen | + | + | + | + | - |
| Prevents bacterial entry | - | - | - | - | + |
| Adherent to nonwounded skin | - | - | + | - | + |
One of the main reasons for using alginates in wound dressings is their haemostatic ability; therefore, alginate dressings can be used for wounds that are bleeding. The coagulation effects of zinc and calcium alginate dressings have been compared with non-alginate dressings. Zinc containing alginates have the best haemostatic ability. Alginate dressings have an important property of gel formation which helps in reducing the pain during removal and changing of the bandage from the wound site. Alginate gels can also be used for the delivery of drugs having low molecular weight. The partially oxidized alginate gels are known to be used for controlled and localized delivery of antineoplastic agents [43, 44].
Alginate dressings have applications in tissue regeneration and bio-engineering fields. Depending on composition, sodium alginate has been identified as a substrate for cell proliferation. This opens up new possibilities for tissue regeneration in skin scaffolds as well [45]. Even though some studies have reported that alginate inhibits keratinocytes migration, Thomas et al., have reported that alginates accelerate healing process by activating macrophages to produce TNF-α which initiates inflammatory signals [46]. In dry condition, alginate dressing removal may damage the newly formed epithelium around the wound. Alginate dressings are suitable for moderate to heavy drainage wounds and not suggested for dry wound, third degree burn wound and severe wounds with exposed bone. Like polymer foam and hydrogel, alginate also needs a secondary dressing to be attached (Table 2) [3].
CONCLUSION
Wound dressings have experienced development to more active dressings which create moist wound environtment. The ideal characteristics of an occlusive dressings are to maintain moist environment, shorten and accelerate wound healing process such as; enhance epidermal migration, promote angiogenesis and connective tissue synthesis, allow gas exchange, improve blood flow to the wound bed and enhance epidermal migration. Moreover, they provide protection against bacterial infection, are non-adherent to the wound providing debridement action, are sterile, non-toxic and non-allergic. Occlusive dressings are not suitable to treat wounds with impaired circulation such as burn ulcers, pressure ulcers, or other heavy trauma.
CONSENT FOR PUBLICATION
Written consent was obtained from the patient for the surgery and the publication of her data.
AVAILABILITY OF DATA AND MATERIALS
Not applicable.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGMENTS
Declared none.


