All published articles of this journal are available on ScienceDirect.
Depression and Quality of Life among Saudi Patients with Vitiligo: A Descriptive Quantitative Review
Abstract
Background:
Vitiligo vulgaris is a disfiguring dermatological disease associated with psychosocial impact and decreased quality of life among its sufferers.
Objective:
To provide an overview of quantitative data on the available literature to evaluate the psychosocial impact of depression among vitiligo vulgaris patients in Saudi Arabia and to assess their quality of life (QoL).
Methods:
We searched MEDLINE (PubMed), ResearchGate, and the Cochrane research database between January 1988 and June 2022 to retrieve all studies that assessed the association between depression and QoL among vitiligo vulgaris patients in Saudi Arabia.
Results:
A total of 11 studies were included—5 studies addressed depression among vitiligo vulgaris patients, and 6 studies addressed QoL. The prevalence of depression among Saudi vitiligo vulgaris patients varies between 37.1% and 54.6%, and the pooled prevalence is 45.81 based on a random effects model. The mean ± standard deviation of the Dermatology Life Quality Index (DLQI) is between 5.64 ± 5.2 and 14.72 ± 5.173. Psychosocial impact is found to be associated with vitiligo vulgaris and an impairment of QoL.
Conclusion:
Depression has common psychosocial symptoms among Saudi vitiligo vulgaris patients and has a negative impact on their QoL.
1. INTRODUCTION
Vitiligo vulgaris is a widely common disfiguring skin disease and has an estimated prevalence of 0.5 to 2% worldwide [1, 2]. It is a multifactorial disorder characterized by depigmentation of the skin and mucosa due to deficiency or inactivation of melanocytes in the basal layer of the epidermis [1-3]. Its exact etiology is not entirely known. It occurs when there is a dysfunction or death of melanocyte cells due to auto-immune disease. The exact triggering factors of vitiligo's immune complex are unknown. However, contemporary evidence suggests a convergence theory as a comprehensive overview of vitiligo vulgaris etiology [4]. The current evidence emphasizes the pivotal involvement of immune cells and their mediators in the pathogenesis of vitiligo vulgaris. Prominent immune cell players encompass T cells, regularity and resident memory T cells, dendritic cells, natural killer cells, and activation of the type 1-IFN pathway [5]. Because vitiligo's appearance greatly impacts body image, it is unsurprising that the psychosocial well-being of patients suffering from it can be greatly impacted [6]. Vitiligo vulgaris is associated with low self-esteem, a tendency to social isolation, self-consciousness, psychological stigmatization, and social discrimination [7-9]. Vitiligo vulgaris patients are vulnerable to depression, anxiety, and social phobia [9, 10]. A growing body of evidence suggests a bidirectional association between depression and vitiligo vulgaris, which can be psychologically devastating [11]. This depression may affect vitiligo vulgaris patients' employment and restrict their social activities. These emotional effects have been reported in several systematic reviews [12, 13]. As sufferers from a chronic and often disfiguring disease, patients with vitiligo vulgaris are also associated with a poor quality of life (QoL) that is especially observed among the Asian and dark-skinned populations, married women, and in patients where vitiligo vulgaris appears on exposed sites [7, 14-16]. Furthermore, since depression and stress can increase systemic inflammation, the interplay between the burden of psychosocial stress and vitiligo vulgaris can worsen disease activity [3]. For these reasons, a conceptualization and management of the psychosocial aspect of vitiligo vulgaris is worth addressing.
In the Kingdom of Saudi Arabia, the prevalence of vitiligo vulgaris varies from 0.5 to 3.5% [17, 18]. Vitiligo vulgaris is found to demoralize the lives of affected patients and their families [11-13]. Considering these somatic morbidities, the psychosocial aspects of vitiligo vulgaris warrant further discussion. Two conceptions are considered in this review. Firstly, the relationship between depression and vitiligo vulgaris among Saudi patients is studied. Secondly, QoL and its effect on vitiligo vulgaris patients is evaluated. Although several studies on the psychosocial well-being of Saudi vitiligo vulgaris patients have been published before, there is no comprehensive understanding of this subject. Therefore, given the clinical importance of this topic, this descriptive quantitative review was carried out to provide a critical reappraisal based on a summary of the results of existing knowledge on depression among vitiligo vulgaris patients in Saudi Arabia. Furthermore, an identification and evaluation of the psychosocial impact of vitiligo vulgaris in a sub-group of Saudi patients tested using different domains of QoL measurements, and an offer of the best evidence of the psychological process for planning therapy for these patients was performed.
2. MATERIALS AND METHODS
2.1. Literature Review
This review was analyzed based on the standard guidelines of Preferred Reporting Items for Systematic Review and Meta-Analyses [19]. A comprehensive online literature review was conducted using search engines such as MEDLINE (PubMed), Research Gate, and the Cochrane database from January 1988 to June 2022. All studies aimed at assessing the association between depression and vitiligo in Saudi Arabia and QoL in vitiligo vulgaris patients were retrieved. As there were no control trials on the subject and due to the heterogeneity of the available studies and lack of common outcome measures (such as odds ratio), we performed a descriptive quantitative review. Thus, we focused on available cross-sectional studies. Our search engine used the following key points: vitiligo vulgaris or skin depigmentation, psychosocial aspects of vitiligo vulgaris (mainly depression), and QoL among Saudi patients.
2.2. Eligibility Criteria
2.2.1. Inclusion Criteria
Articles were included in the study if they met the following criteria: (1) Studies that had a baseline description of the prevalence of depression among Saudi vitiligo vulgaris patients. (2) Studies that examined the QoL among these patients. (3) Studies that were published in the English language. (4) Studies that were considered to be medium- or high-quality based on the Newcastle–Ottawa Quality Assessment Scale [20].
2.2.2. Exclusion Criteria
Our exclusion criteria were as follows: (1) Cohort studies that did not state the prevalence of depression among Saudi vitiligo vulgaris patients. (2) Studies with insufficient data as well as conference abstracts, case reports, case studies, review articles, or commentaries. (3) Studies that were considered to be of low quality based on the Newcastle–Ottawa Quality Assessment Scale.
2.3. Data Extraction
Two authors (FZ and AZ) used standard data extraction based on predefined inclusion and exclusion criteria and independently performed the study selection. Any dispute that arose was resolved through joint discussion. The following data was extracted from the selected studies: name of author, place and year of publication, study design, number of participants, gender, age, prevalence of depression, and QoL among vitiligo vulgaris patients. Due to the heterogeneity of study designs, the authors sought cross-sectional studies. Hence, the Newcastle–Ottawa Quality Assessment Scale was used to assess the comparability and quality of the chosen studies. The quality of the articles was graded as follows: low: <4, moderate: 4–6, and high: >6 [20]. A grade less than 4 was considered insufficient (Tables 1 and 2).
2.4. Quality and Risk of Bias
In this review, we encountered several methodological problems. There were no randomized control trials of the subjects, and neither were their case controls or cohort studies. The only available study design was cross-sectional.
Data Analysis: There was an inherent flaw in the studies included in this review. Hence, a complete meta-analysis was not possible due to inadequate reporting of similar measures (OR). Therefore, a quantitative review with descriptive meta-analysis was carried out. The Meta-analysis in this systematic review was carried out to estimate the combined (pooled) estimate of proportion (prevalence) of depression in vitiligo vulgaris patients. Forest plots were used to show graphically the pooled prevalence (using both fixed and random effect models) of studies included in the meta-analysis. The Meta-analysis was carried out using MedCalc for Windows version 15.0 (MedCalc Software, Ostend, Belgium).
| Series | Authors/Refs. | Year | Type of Study | Region | N | No. of Depress Pts | Participants | Tools to Measure Depression | Outcomes Measured | NOS Score |
| 1 | Alhetheli I et al. [21] | 2021 | Cross sectional | Central, Saudi Arabia | 173 | 93 | Mean age- 37.2 ±8.6 BDI II Median Score: Mild depression (25.4±3.3), Moderate Depression (36.1±2.8) 45.1% have mild depression while 8.7% have moderate depression - the sense of being unattractive is the most significant cause of depression (AUC: 0.639±0.05 ; CI: 0.530-0.748, p=0.02). |
BDI II | Assess the severity of current depressive symptoms | 4 |
| 2 | Almomani et al. [22] | 2015 | Cross sectional | Central, Saudi Arabia | 234 | 64 | Mean age- 30.8±11.23 Mean score HAD for depression: 490±4.06 Females had higher score in HAD for depression (57.8%) compared to males but not significant, meanwhile 60% of low income patients have higher depression score compared to those with higher income, p=0.028. |
HADS | Assess depression and anxiety | 5 |
| 3 | Alharbi [23] | 2020 | Cross sectional | Central, Saudi Arabia | 308 | 168 | Mean age= 27±14.5 As per BDI scale 54.5% has depressive symptoms; 52.4%- mild depression; 33.3%-moderate depression 14.3%- severe depression. The prevalence of depression is higher and significantly associated with age (children & adolescent (68.3%)(p=0.003) ; marital status (singles-61.2%) (p,0.001); educational level (lower than high school-64.9%)(p<0.001); shorter duration of the disease (66.7%, ,p<0.001). |
BDI | Assess depression | 5 |
| 4 | Alsawaf et al. [24] | 2020 | Cross -sectional | Western, Saudi Arabia | 58 | 34 | Mean age: 27±8.564 As per DASS 42 scale; mean score: 22.328±11.106; extremely severe depressed (34.5%), severe depressed (24.1%), mildly depressed (17.2%) Significant association of depression with age, higher in patients less than 30 years old (p=0.001); sex (female (27.889±11.517, p=0.012) ; marital status (singles (28.303±8.907, p=0.001); educational level ( post graduate (22.938±8.48, p=0.001); duration of the disease (< 5 years (33.842±5.728, p=0.001). |
DASS 42 | Assess depression, anxiety, and stress | 5 |
| 5 | Shatla et al. [25] | 2016 | Cross-sectional | Western, Saudi Arabia | 132 | 49 | Mean age -30.1± 13.2 Met HADS ≥ 11 (37.1%) criteria for depression; 13.8% for depression alone. Significant association of risk for depression: age (<25 yrs, p=0.03); marital status (single (p=0.013; affected area (exposed, p=0.035); perception of poor personal control (p=0.001) and illness consequences (p=0.003). |
HADS | Assess depression and anxiety | 4 |
Table 2.
| Series | Authors/Refs. | Year | Type of Study | Region | N | Participants | Tools to Measure Quality of Life | Outcomes Measured | NOS Score |
| 1 | Almomani et al. [22] | 2015 | Cross sectional | Central, Saudi Arabia | 234 | Mean age- 30.8±11.23 Mean score DLQI – 8.72±6.09 33.8% of patients felt that having vitiligo have large/ extremely large social effect on their lives; significant association on age (<30; 62%, p=0.00); sex(female , 67.1%, p=0.04), income capacity (low income, 63.3%, p=0.00), involvement of the neck p<0.028); and >50% of body surface involvement (p=0.013). |
DLQI | to measure the health-related quality of life of adult patients suffering from vitiligo | 5 |
| 2 | Al Robaee et al. [15] | 2007 | Cross sectional | Central, Saudi Arabia | 109 | Mean age- 26.94±9.73 Mean score DLQI – 14.72±5.173 Married males have higher DLQI score compared to married females (p<0.05), 77.9% of patients feels much a lot of embarrassment because of vitiligo especially on females (<0.005); on choosing clothes to wear (p,0.05), impairment on social life (61%, p<0.001); relationships (p<0.001) DLQI score is higher in patients with generalized vitiligo (16.11±4.96, p,0.05) Family history of vitiligo is significant factor in 50% females vs 27.5% males (p<0.005). |
DLQI | to measure the health-related quality of life of adult patients suffering from vitiligo | 5 |
| 3 | Al-mubarak et al. [26] | 2011 | Cross-sectional | Central, Saudi Arabia | 260 | Age: Range Male-19-64 (±9.8), Female 18-53 ((±7.6) In the dimension of QoL Scale used significant difference between male and female QoL Scale 11.1 vs 23.92 (p<0.001), mean score between married and divorced 3.24 vs 7.75 (p<0.05). Master degree holder had a lower QoL score compared with others 1.25 vs 10.7,9.7, and 5.6 The age group 21-30 significant difference (p<0.05). compared with other age groups The QOL scale was higher in those with exposed vs non exposed lesions 5.0 vs3.4 (p<0.05). |
Customized QoL Questionnaire | to measure the health-related quality of life of adult patients suffering from vitiligo | 4 |
| 4 | Al Shammari et al. [16] | 2021 | Cross sectional | Central, Saudi Arabia | 253 | Mean DLQI score- 5.64±5.2 SD Male (4.0), Female (5.0) (p=0.09) Median DLQI score higher in married and with progressive vitiligo (p<0.05), no difference in age, gender, education level and occupation 75% with vitiligo areas that have profound impairment effect on DLQI on exposed body surface areas like face, hand and feet. |
DLQI | to measure the health-related quality of life of adult patients suffering from vitiligo | 4 |
| 5 | Al-Shobaili et al. [27] | 2015 | Cross sectional | All Regions, Saudi Arabia | 48 | Mean Age: 29±10.9 Mean QoL score- before therapy 10.6±4.3 QoL Score statistically significant after excimer laser treatment (p<.001) QoL- Significant improvement in 5 domains of QoL after excimer laser treatment (symptoms & feeling; daily activities; leisure; work & school; and personal relationships (p<.001) Female have longer disease duration, lesions on the face and neck have significant improvement in QoL score after excimer laser treatment (p<.01). |
Customized QoL Questionnaire | to measure the health-related quality of life of adult patients suffering from vitiligo | 4 |
| 6 | Alhetheli et al. [21] | 2021 | Cross sectional | Central, Saudi Arabia | 173 | Mean age- 37.2 ±8.6 Median Score (VIS-22): 28 (IQR: 24;33) Age has an inverse relationship but significant factor in higher VIS-22 score (p<0.001) as well as the duration of disease (p<0.001). |
VIS-22 | to measure the health-related quality of life of adult patients suffering from vitiligo | 4 |
3. RESULTS
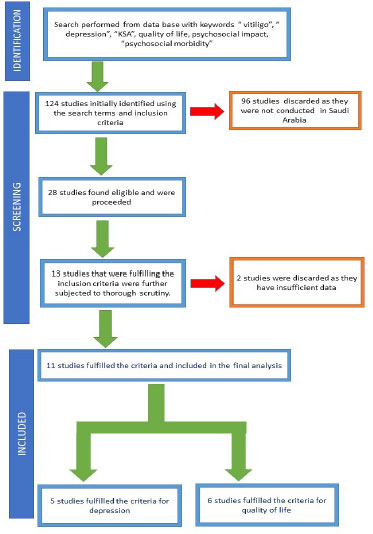
The initial database generated a preliminary 124 articles on patients with vitiligo vulgaris. After screening the title and abstract, 96 articles were discarded as they were review articles and were not done in Saudi Arabia. Therefore, 28 articles were found eligible. Finally, 13 articles were found to fulfil the inclusion criteria. Two other studies were also discarded because of insufficient data. The remaining (n = 11) articles fulfilled the inclusion criteria and were included in the final analysis. (Fig. 1) All the included studies were cross-sectional. Of the included studies, 5 examined the association between vitiligo vulgaris and depression (Table 1) [21-25], while 6 studies examined the association between vitiligo vulgaris and QoL (Table 2) [15, 16, 21, 26, 27] Inter-rater agreement between the 2 researchers was 96%. There was considerable methodological variability, and the data extracted from different studies was heterogeneous. Most of the studies used BDI and HADS scales to assess depression.
3.1. Prevalence of Depression among Saudi Patients with Vitiligo
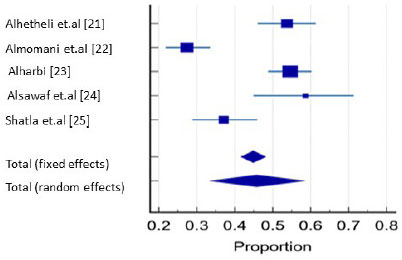
All studies did not mention whether their subjects had been previously diagnosed with depression. Rather, we examined whether the depressive symptoms mentioned in a questionnaire were answered. The prevalence of depression is based on a summary of articles, and it varied in different studies and ranged between HAD score = 27.3% by Almomani et al. [22] and BDI-II = 54.6% by Alharbi et al. [23]: both of these studies were conducted in Riyadh. Studies done in Western regions of Saudi Arabia reported DASS 22 = 58.6% (Mecca) [24] and HAD = 37.1% (Al-Madinah) [25]. The pooled prevalence of depression was 45.81% based on random effects (Table 3, Fig. 2). The variation in these studies may suggest a methodical bias and the use of different assessment scales, as there was statistically significant heterogeneity (Q = 56.02 and p <0.001) with 92% inconsistency across the studies included. Nevertheless, the findings of this review should be considered with caution.
3.2. Factors associated with Depression in Vitiligo Vulgaris Patients
The results of the studies showed that vitiligo vulgaris patients in a lower age group (<30 years old) are more prone to depression than vitiligo vulgaris patients in older age groups [23-25]. Furthermore, depression is more prevalent among females than it is among males [22, 24]. When we probed the results of the articles reviewed, we found patients perceive hands and exposed areas as the most impactful part of the body: they had an OR of 3.9 (1.1–14.2) and a p-value of 0.035 [25]. Depression was associated with a duration of disease of less than 5 years in different studies (P value = 0.001) [24, 25]. Also, Alharbi et al. [23], Shatla et al. [25], and Alsawaf [24] reported that the association of depression with marital status was significantly high among single patients (p = 0.006, p < 0.035, and p < 0.001), respectively, while others found no significant difference between single and married patients [22]. Most of the articles suggest that vitiligo vulgaris patients with high education are likely to have depression (P < 0.001, p = 0.037) [22, 24]. Alharbi et al. [23] reported that depression is high among patients with a low educational level (p < 0.001). The employment status of vitiligo vulgaris patients has no significant association with being depressed [22, 24, 25]. Pooled odds ratios could not be arrived at due to the lack of this outcome measure across the studies for any factors associated with depression in vitiligo vulgaris patients.
| Study/Refs. |
Sample size |
Proportion (%) |
95% CI | Weight (%) | |
|---|---|---|---|---|---|
| Fixed | Random | ||||
| Alhetheli et al. [21] | 173 | 53.757 | 46.030 to 61.354 | 19.12 | 20.37 |
| Almomani et al. [22] | 234 | 27.350 | 21.745 to 33.539 | 25.82 | 20.75 |
| Alharbi [23] | 308 | 54.545 | 48.801 to 60.201 | 33.96 | 21.02 |
| Alsawaf et al [24] | 58 | 58.621 | 44.927 to 71.404 | 6.48 | 17.91 |
| Shatla et al. [25] | 132 | 37.121 | 28.877 to 45.957 | 14.62 | 19.94 |
| Total (fixed effects) | 905 | 44.885 | 41.620 to 48.184 | 100.00 | 100.00 |
| Total (random effects) | 905 | 45.812 | 33.503 to 58.386 | 100.00 | 100.00 |
3.3. Vitiligo and Quality of Life
Six studies quantified the quality of life in 1,077 patients with vitiligo vulgaris [15, 16, 21, 22, 26]. Total DLQI score was used in 3 studies [15, 23] and interpreted as follows: 0–1 = no effect on a patient's life, 2–5 = a modest effect, 6–10 = a moderate effect, 11–20 = a large effect, and 21–30 = an extremely large effect [28]. Two studies used customized QoL instruments [26, 27], and one used VIS-22 [21]. The mean level of the DLQI score varied in different studies: Al Robaee et al. [15], with a mean of 14.72 ± 5.173, had a high score, while Alshammari et al. [16], with a mean of 5.64 ± 5.2, had a low score. The DLQI score was significantly high among female patients with vitiligo vulgaris in 3 studies (p = 0.04, p < 0.005, and p < 0.001), respectively [15, 22, 26]. The rest of the studies showed no significant difference between genders. The score was significantly higher among the married than it was among singles in 2 studies [15, 16] (p < 0.001 and p < 0.05, respectively), while the study by Al-Mubarak et al. [26] showed that the mean score was higher in singles compared to married patients: 5.09 versus 3.24, respectively; however, among the divorced, the score was 7.75 (p < 0.05). The DLQI mean score in patients with exposed lesions was higher than it was among patients with non-exposed lesions (p < .028 and p < 0.05) [22, 26], respectively. Female patients, compared with males, feel more embarrassment (p < 0.005) in choosing clothes to wear (p = 0.05), concerning impairment in social life (61%; p < 0.001), and in relationships (p < 0.001) [15]. One study measured the intention to commit suicide and found that the DLQI score was higher in those who attempted suicide, 21.67 ± 4.93, compared to those who were non-suicidal [22].
4. DISCUSSION
This review was conducted to understand the adverse effect of vitiligo vulgaris on the psychological well-being of Saudi patients. Despite not being life-threatening, the uncertainty of the clinical course of the disease and a lack of effective therapy causes vitiligo vulgaris to have a profound effect on the psychosocial being of patients. Therefore, we aimed to update and expand previous reports on the subject and quantify the level of depression and QoL among these patients. The outcome was inconsistent among studies owing to substantial variability of the results. This review suggests that Saudi patients with vitiligo vulgaris experience a significant disease-related burden and self-perceived depression. In the present study, the pooled data showed that the prevalence of depression is higher among Saudi patients with vitiligo vulgaris compared with average reported data on depression in the general population but is consistent with many international reports [13, 29, 30]. Several systematic reviews and a meta-analysis of the prevalence of depression among vitiligo vulgaris patients yielded divergent results: 12.2% to 59% [13, 31-33]. This variation is attributed to diverse ethnic backgrounds, geographical locations, and different assessment tools. In the current review, a young age (<30 yrs), female sex, and a short duration of the disease (less than 5 yrs) were found to be associated with perceived depression. In addition, marital status and low educational levels also offer protection, while the presence of vitiligo vulgaris in an exposed area often has an additional impact on depression. These results are in concurrence with previous reports [12, 32]. Depression is a significant impact factor in the long-term health of patients with chronic diseases, including vitiligo vulgaris, and recent data indicates that there is an increased risk of mortality among patients with depression [32-34]. Moreover, vitiligo vulgaris may increase depression, and, in turn, depression could exacerbate vitiligo [3]. The pathogenesis of depression in vitiligo vulgaris patients is unknown. However, the increased activity of hypothalamic–pituitary access with an increased level of catecholamines during chronic stress activates α receptors in skin arterioles, causing vasoconstriction and leading to microcirculation disturbance and epidermal and dermal hypoxia. As a result, the generation of oxidative stress results in cellular stress and the eventual destruction of melanocytes [35, 36]. Furthermore, there is an assumption that H2O2-mediated oxidation of acetylcholine esterase causes substantial oxidative stress in vitiligo vulgaris [37]. This assumption may underlie the theory behind the prevalence of depression among vitiligo vulgaris patients. Changes in neurotransmitters have been postulated as the reason behind using antidepressant agents such as amitriptyline, which has significant anticholinergic action and mild norepinephrine reuptake blockage in patients with vitiligo vulgaris and depression [38].
Data from different studies, including ours, show an impairment of the QoL among vitiligo vulgaris patients with gender differences, and the QoL score among vitiligo vulgaris patients is generally lower among females than among males [39-43]. This may be because females are vulnerable to experiencing negative emotions regarding their aesthetics, including anxiety, depression, and embarrassment from the stigma of feeling different because of great societal pressures on the female physical appearance [8, 43, 44].
The young are particularly psychologically affected by vitiligo vulgaris and have an overall great impairment of QoL since their age group is especially conscious of physical appearances and social acceptance. The young may view this disease as detrimental to their self-esteem and hindering their chance of finding a life partner [45, 46]. The findings of this review are also consistent with previous studies from Iran and Korea, which showed that QoL is poor among young age groups [42, 47]. Although this review showed that being married resulted in a higher QoL score compared to being single, there are conflicting results between married males and married females. Moreover, the effect of vitiligo vulgaris on the QoL of the married subjects is not as profound in the long run as it is in a single person with vitiligo vulgaris. Marriage is thought to profoundly affect the overall well-being of a vitiligo vulgaris patient as their significant other can offer emotional, physical, and financial support [48]. Unlike singles, the psychological burden of the disease is intense, as having vitiligo vulgaris decreases the chances of intimate relationships and marriage [49].
The impact of the affected body parts- especially exposed areas such as the face, hands, and feet and lesions on a large part of the body- significantly affects the patient's QoL. The result from this review can be compared to previous studies by Bae et al. [50] and Benny et al. [41]. Vitiligo vulgaris patients may view themselves as flawed and hideous, which may negatively affect their psychological condition as well as their social and professional relationships. Furthermore, this negative perception about oneself can be either a driving force to seek treatment or to hide the visible areas affected using makeup or clothing [32, 39, 51]
5. LIMITATION OF THE STUDY
Our study has methodological shortcomings. Firstly, the reviewed papers were cross-sectional research, which can impede a strong conclusion due to their inherent flaws. Secondly, there may be selection bias as all subjects were recruited from dermatology clinics; this may be misleading because patients who suffer from few psychological complaints are less likely to seek medical advice. Finally, because different instruments were used to quantify depression in different study groups, comparing these studies is imprecise.
CONCLUSION
This review provides a comprehensive overview of existing quantitative data on the impact of vitiligo vulgaris on the mental health of Saudi patients. Vitiligo vulgaris has a substantial negative effect on the QoL of these patients. It is, therefore, important that depressive illness in patients with vitiligo vulgaris is addressed by dermatologists. Screening these patients for depression may help treatment outcomes. Future research should concentrate on high-risk groups in need of psychiatric referral. Owing to the heterogeneity of the results, the data should be interpreted with caution.
LIST OF ABBREVIATIONS
| QoL | = Quality of life |
| BDI | = Beck Depression Inventory |
| BDI-II | = Beck Depression Inventory-II |
| DASS 22 | = Depression Anxiety and Stress Scale 22 |
| DLQI | = Dermatology Life Quality Index |
CONSENT FOR PUBLICATION
Not applicable.
STANDARDS OF REPORTING
PRISMA guidelines and methodology were followed.
AVAILABILITY OF DATA AND MATERIALS
The data that support the findings of this study are available from the corresponding author, [F.A], upon reasonable request.
FUNDING
None.
CONFLICT OF INTEREST
The authors declare no conflict of interest, financial or otherwise.
ACKNOWLEDGEMENTS
Declared none.
SUPPLEMENTARY MATERIAL
PRISMA checklist is available as supplementary material on the publisher's website along with the published article.
Supplementary material is available on the publisher's website along with the published article.


